Diversity: Improved Sample Prep Speed and Accuracy Depend Upon Workflow
A comprehensive review of sample preparation would require a multivolume work. Beyond that, covering the possible number of workflows given the hundreds of potential “unit operations” would fill an encyclopedia, especially when solids (rock, soil, animal tissue) are considered.
A comprehensive review of sample preparation would require a multivolume work. Beyond that, covering the possible number of workflows given the hundreds of potential “unit operations” would fill an encyclopedia, especially when solids (rock, soil, animal tissue) are considered.
ENVIRONMENTAL
While the major market share for automated sample prep lies in the life sciences, which operate with very small samples, many advantages are achievable outside biology, where larger samples rule—albeit perhaps not always at the same level of automation. For example, Horizon Technologies specializes in automating the extraction of large volumes of aqueous samples, principally for environmental analysis. EPA methods call for collecting up to two liters of water when testing for various contaminants. Due to sometimes very low concentrations of analytes, some tests demand collection of even larger volumes.
Horizon’s SPE-DEX 4790 automated extractor system accepts sample bottles without manipulation. Samples are initially filtered to remove particulates and then adsorbed onto a solid phase extraction disk. The system then extracts analytes from the disk. Additional concentration steps, which render the sample suitable for LC or GC analysis, are also automated. Up to eight extractors may be run simultaneously through a single computer controller.
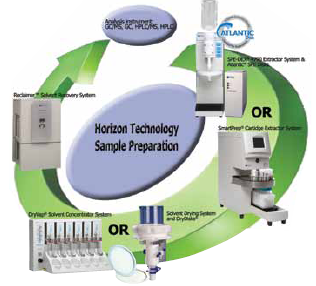 Sample flow through automated extraction, drying and evaporation/concentration processes.“Most testing labs are still using ‘bucket chemistry’ to process large-volume samples,” says Zoe Grosser. “Conventional extraction, drying, and concentration from volumes that large can take anywhere from one to three hours, depending on the sample’s complexity and the analytes of interest. Here it takes just 30 minutes per extraction, and it’s a walkaway operation.”
Sample flow through automated extraction, drying and evaporation/concentration processes.“Most testing labs are still using ‘bucket chemistry’ to process large-volume samples,” says Zoe Grosser. “Conventional extraction, drying, and concentration from volumes that large can take anywhere from one to three hours, depending on the sample’s complexity and the analytes of interest. Here it takes just 30 minutes per extraction, and it’s a walkaway operation.”
Grosser outlines the usual list of benefits: increased user productivity, decreased solvent consumption, fewer errors, greater consistency, lower labor costs, fewer re-extractions, freeing workers for other tasks, reduced glassware purchase and cleaning (the extractor cleans sample bottles after emptying them), and improved external ROI. “Labs are able to convert more samples per unit time, reduce customer support costs, and improve brand value of their services by informing customers of their automation capabilities. Automation sells.”
GENOMICS
In high-throughput genomics, assays are lengthy, consume many thousands of dollars in reagents, and are limited by tissue availability and quality. Sample preparation can consume several days. “Researchers don’t know how the tests are going until the very end,” says PerkinElmer’s Jeremy Lambert. PerkinElmer’s key market areas are high-throughput genomics (mostly next-generation sequencing assays) and biotherapeutics discovery.
Kits originally developed for high-throughput genomics were designed for manual lab processing: An operator with a multichannel pipette might process up to 15 samples at a time through a workflow several days long that is highly demanding of hands-on time. “Transforming bench-worthy workflows to automation while maintaining quality requires significant optimization,” Lambert says.
Genomics labs tend to conduct similar protocols that rapidly evolve due to improvements in techniques, methods, reagents, and scientific understanding. Vendors introduce new protocols approximately every three to six months. To prevent “method creep,” some vendors employ single, fixed configurations for much of their liquid handling equipment, so new methods can be deployed to the installed base very quickly.
 HPLC Autosamplers / Flexar / PerkinElmer / www.perkinelmer.com PerkinElmer’s other specialty, biotherapeutics, involves a completely different set of tools. Where genomics chews up DNA and RNA to determine their molecular sequences, cell-derived proteins are analyzed for size, purity, charge, activity, and other critical quality attributes, usually by high-performance liquid chromatography and LC-mass spectrometry. Sample preparation is critical to ensure that analytical tests provide the right information in a competitive environment where one-day delays in market entry are punished. “In protein labs researchers are highly trained biochemists who need to focus not on sample preparation but on more challenging tasks, such as interpreting assay results. Automation frees them to do so.”
HPLC Autosamplers / Flexar / PerkinElmer / www.perkinelmer.com PerkinElmer’s other specialty, biotherapeutics, involves a completely different set of tools. Where genomics chews up DNA and RNA to determine their molecular sequences, cell-derived proteins are analyzed for size, purity, charge, activity, and other critical quality attributes, usually by high-performance liquid chromatography and LC-mass spectrometry. Sample preparation is critical to ensure that analytical tests provide the right information in a competitive environment where one-day delays in market entry are punished. “In protein labs researchers are highly trained biochemists who need to focus not on sample preparation but on more challenging tasks, such as interpreting assay results. Automation frees them to do so.”
MICROFLUIDICS : HOW LOW CAN YOU GO?
DNA extraction from cell cultures for subsequent amplification is an example of a complex sample prep that is often the rate-limiting step of a microbiological workflow. DNA is separated on microscale chromatography columns in a process that requires pipetting, dispensing, and pressurizing to elute the sample through a Q-Sepharose column. All three functions are available in automated formats but typically as separate instruments. A group at the Fraunhofer Center for Manufacturing Innovation at Boston University (Brookline, MA) has developed a prototype device, the Tripette, which combines the three essential functions through a six-by-six-inch microchanneled polycarbonate manifold that sits above a standard microtiter plate. Backing up the Tripette are standard fluid delivery and pressure systems. “The innovation is not the services behind the scenes but the manifold itself,” says Alexis Sauer-Budge, Ph.D., one of the Tripette’s inventors. Fraunhofer is interested in codevelopment and collaboration on the Tripette and other sample prep technologies found at the website http://bit.ly/RImeSs.
In contrast to the almost macroscale Tripette, Akonni Biosystems (Frederick, MD) has built an integrated, microfluidic-controlled microarray platform for sample prep and analysis of genetic mutations known as single-nucleotide polymorphisms (SNPs). Targeted at forensic applications, the microarray incorporates Akonni’s gel drop microarray technology, a thermal cycler, a reader, and a cartridge dock in a single-use “sample to analysis” microchanneled platform.
A WORD ON SPEED
 Automated Extraction System / SPE-DEX 4790 Horizon Technology / www.horizontechinc.com A misconception regarding automation in general and automated sample preparation in particular is that it improves throughput. This line of reasoning suggests a barrier—some number of samples per day—before which automation is unwarranted and beyond which it becomes sensible.
Automated Extraction System / SPE-DEX 4790 Horizon Technology / www.horizontechinc.com A misconception regarding automation in general and automated sample preparation in particular is that it improves throughput. This line of reasoning suggests a barrier—some number of samples per day—before which automation is unwarranted and beyond which it becomes sensible.
According to Beckman’s Mehul Vora, the “throughput” argument collapses in the face of the realities of quality and workflow. “Intuition says if you’re dealing with just a few samples, you might just as well do them by hand because it’s faster than setting up a machine. But our customers tell us otherwise.” Automation’s major advantages over manual sample prep are fourfold: avoiding human error, eliminating operator-related contamination, enhancing data quality, and maintaining consistent results.
Manual sample prep offers the illusion of control over the process. The question is what is controlling whom? A prep may take five steps that are mostly walkaway; for example, weighing, dissolving, filtering, adding reagent, and heating. Each operation may require only ten or 15 minutes of attention, but the process keeps drawing the scientist or technician back from other work. Five samples can easily keep a lab worker tied to prep work for half a day, even more if operations require monitoring. “And if the number is 55 samples, the value of automation becomes even more clear,” Vora says.
Automation does not necessarily speed up prep work. If one includes time to set up the method, absolute throughput—the number of samples processed per day—may hardly be affected. If productivity is measured by what a typical worker can accomplish in a shift, however, the equation changes. Now, instead of babysitting samples and individual operations, a worker may spend half an hour setting up reagents and selecting a method, occasionally moving plates, taking a few peeks during the day. In other words, the worker is freed to perform less repetitive, more challenging tasks. “Even low-throughput labs can save value-added time.”
