Scientists Locate Nerve Cells That Enable Fruit Flies to Escape Danger
Findings offer clues into comparable survival strategies in people
A fruit fly performs the bend-and-roll escape behavioral sequence. In this study, scientists identified a class of nerve cells, called down-and-back neurons, that initiate this behavior in response to dangerous stimuli. Video credit: Anita Burgos/Grueber Lab/Columbia's Zuckerman Institute
Columbia University researchers have identified the nerve cells that initiate a fly's escape response: that complex series of movements in which an animal senses, and quickly maneuvers away from, something harmful such as high heat. These results, based on observations in fruit fly larvae, provide a window into a survival mechanism so important that it has persisted across evolutionary time, and today exists in virtually all animals—including in people. They also lend insight into conditions characterized by dysfunctions in this response, such as allodynia, in which gentle touch triggers the same reaction as exposure to something harmful.
The study was published this week in the journal eLife.
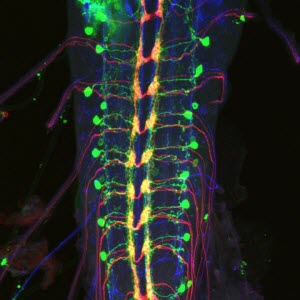 Down-and-back neurons (green) and nociceptive sensory neurons (red) in the ventral nerve cord of a fruit fly. Nerve bundles (blue) serve as an anatomical landmark.Image credit: Anita Burgos/Grueber Lab/Columbia's Zuckerman Institute"Protecting ourselves from danger is a critical tool for survival that we employ all the time, whether it's yanking our hand away from a hot stovetop, or ducking our head to avoid a low ceiling," said neuroscientist Wesley Grueber, PhD, a principal investigator at Columbia's Mortimer B. Zuckerman Mind Brain Behavior Institute and the paper's senior author.
Down-and-back neurons (green) and nociceptive sensory neurons (red) in the ventral nerve cord of a fruit fly. Nerve bundles (blue) serve as an anatomical landmark.Image credit: Anita Burgos/Grueber Lab/Columbia's Zuckerman Institute"Protecting ourselves from danger is a critical tool for survival that we employ all the time, whether it's yanking our hand away from a hot stovetop, or ducking our head to avoid a low ceiling," said neuroscientist Wesley Grueber, PhD, a principal investigator at Columbia's Mortimer B. Zuckerman Mind Brain Behavior Institute and the paper's senior author.
"These movements are quick, but complex, so teasing apart the brain activity that guides them has long proven difficult," he continued. "With today's findings, we can now confirm which nerve cells in flies orchestrate this process, bringing us closer to understanding the brain mechanisms that guide this essential behavior that evolved to keep us safe."
In broad terms, the steps that dictate a fly's escape can be divided into two parts: first the animal detects danger and then it reacts to it. Previous work found that specialized nerve cells in the animal's sensory system, known as nociceptive neurons, act as detectors, switching on in the presence of danger and alerting flies to make their escape, first by bending themselves into a c-shape and then rolling out of harm's way.
"The question for us then became: How do these nociceptive neurons send information back to the brain, and how does this result in the animal's escape?" asked Anita Burgos, PhD, a postdoctoral researcher in the Grueber Lab and the paper's first author.
To find out, the team used a revolutionary technique known as electron microscopy (EM) reconstruction. EM reconstruction allows researchers to visualize the pathways that link different neurons—like tracing the route between two cities on a road map.
By following the route nociceptive neurons took toward the brain, the researchers saw that the neuronal branches all terminated in the same region of the ventral nerve cord (the fly equivalent of our spinal cord). Upon closer inspection, the team found that this region was largely home to one type of cell: down-and-back neurons, so named because of their curved shape.
This discovery offered strong evidence that down-and-back neurons may be the key to the fly's escape response. To confirm this, the researchers used genetics to manipulate the activity of down-and-back neurons, and observed their resulting behavior.
"When we switched on the down-and-back neurons, the flies performed the classic bend-and-roll escape even in the absence of harm," said Dr. Burgos. "But when we silenced those same neurons, the animals could still sense danger, but couldn't escape it. Their bodies wouldn't bend correctly; and were also unable to roll away. Somehow, down-and-back neurons were driving both of these behaviors—bending and rolling—almost simultaneously."
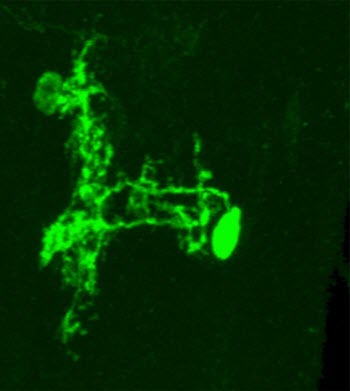 A down-and-back neuron (green), so named because of its curved shape.Image credit: Anita Burgos/Grueber Lab/Columbia's Zuckerman InstituteFurther experiments revealed how it worked. Upon receiving a danger signal from the nociceptive neurons, down-and-back neurons sent two sets of instructions to the animals' muscles—one that initiated the bend, and a second that initiated the roll. At first, it seemed counterintuitive for the same set of neurons to be in charge of driving two different types of movement.
A down-and-back neuron (green), so named because of its curved shape.Image credit: Anita Burgos/Grueber Lab/Columbia's Zuckerman InstituteFurther experiments revealed how it worked. Upon receiving a danger signal from the nociceptive neurons, down-and-back neurons sent two sets of instructions to the animals' muscles—one that initiated the bend, and a second that initiated the roll. At first, it seemed counterintuitive for the same set of neurons to be in charge of driving two different types of movement.
"But in fact, the cellular activity we witnessed is an evolutionary solution for accomplishing the virtually limitless permutations of behaviors that animals can perform—a way for the brain to reuse the same neurons to perform different, but related, duties," said Dr. Grueber, who is also associate professor of physiology & cellular biophysics and neuroscience at Columbia University Irving Medical Center.
"Even in a very simple organism such as the fly, we're just beginning to understand how distinct movements are strung together into complex sequences," added Dr. Burgos, "This new study is a significant step towards understanding any complex behavior that is made up of simpler actions that are linked together in sequence, such as human speech."
The detailed mapping of brain circuits in this study may also provide insights into the mechanisms that guide the sensing abilities of other species, including people.
"This could ultimately be important for understanding how touch and pain are sensed separately, and how the two senses may become conflated in conditions such as allodynia, in which even the gentlest touch is interpreted as painful or dangerous," said Dr. Grueber.
