Scientists Provide New Insights into Gene Regulation
Researchers at the University of Leicester have solved the three-dimensional structure of a gene repression complex that is known to play a role in cancer
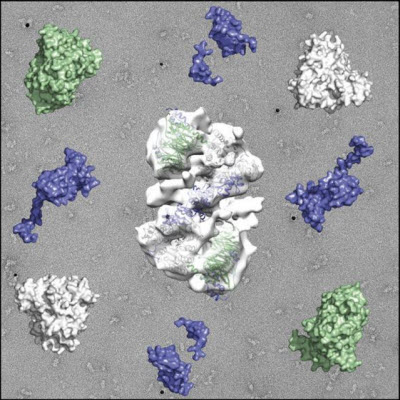 This is an image of the NuRD complex.Image Courtesy of: University of LeicesterA team of researchers led by the University of Leicester has shed new light on how the regulation machinery that controls gene expression works by characterising a complex known as the NuRD complex.
This is an image of the NuRD complex.Image Courtesy of: University of LeicesterA team of researchers led by the University of Leicester has shed new light on how the regulation machinery that controls gene expression works by characterising a complex known as the NuRD complex.
The study, led by John Schwabe, Professor of Structural Biology and Head of Department of Molecular and Cell Biology at the University of Leicester and published in the journal eLIFE, focuses on three protein components which make up the core of the NuRD complex: MTA1, RBBP4 and HDAC1.
The function of the NuRD complex is to control the amount of protein made by the cell—a process called gene regulation. Unregulated protein expression disrupts the equilibrium within a healthy cell and can lead to abnormal cell division and tumour growth.
The NuRD complex components HDAC1 and MTA1 have both been shown to be present in high levels in certain cancers. A clearer picture of how these proteins interact may help to develop strategies to reduce the activity of the complex and bring the cell back into equilibrium.
Related Article: Study Uncovers Inherited Genetic Susceptibility Across 12 Cancer Types
By examining the relationship between the three proteins, the team was able to show unprecedented detail about the 3D structure of the NuRD complex as well as to characterise the molecular nature of the 'extensive interface' between MTA1 and RBBP4.
Professor Schwabe said: "This work is hugely exciting as the images from the electron microscopy provide a direct 3D picture of a repression complex and from this we have been able to identify one of the recruitment mechanisms for targeting to specific genes. The 3D structure of the NuRD complex shows that the interaction between components is more extensive than was previously known.
"This study opens up the possibility of solving the structure of the entire molecular machine and raises the challenge of exploring how the additional subunits fine-tune gene regulation.
"This study has only been possible through a fruitful collaboration between groups at the University of Leicester and the University of Warwick with support from the Diamond Light Source near Oxford. We are delighted to have been able to share resources and expertise in generating these results.
The findings paint a clearer picture about how the complex is assembled and the way in which it interacts with the proteins that package the human genome in the cell. A deeper knowledge of how these complexes are recruited to genes will help to design treatments to combat aberrant gene activity.
Dr Christopher Millard from the University of Leicester Department of Molecular and Cell Biology added: "In the future, an understanding the exact mechanism by which the NuRD complex assembles and functions could make it an attractive target for developing novel and specific anti-cancer therapeutics and treatment for a range of genetic diseases."
###
The research was funded primarily by the Wellcome Trust and the Biotechnology and Biological Sciences Research Council (BBSRC) with support from the Royal Society.
The paper 'The structure of the core NuRD repression complex provides insights into its interaction with chromatin' is published in the journal eLIFE is available at: DOI: dx.doi.org/10.7554/eLife.13941
