Problem: Assays using immortalized cell lines and plastic labware have been used for decades as an in-vitro mimic of how a putative drug may work in the human body. The last decade has seen attempts to improve the physiological relevance of these assays by the incorporation of human primary cells that better reflect human biology both from a drug efficacy and toxicity perspective. At issue with the use of primary cells is the relative difficulty in tissue culturing and maintenance for long-term studies relative to immortalized cell lines.
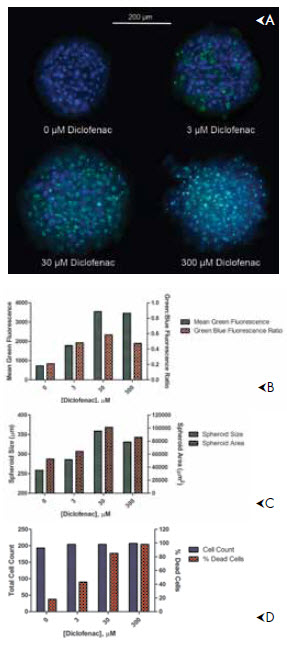
Solution: 3D cell culture methods that involve cell aggregation either by self-assembly into spheroids or the use of scaffolds to build tissue-like structures can enable long term experiments spanning a week or more. Peter Banks and Brad Larson of BioTek have shown that human hepatocytes demonstrate excellent plasma membrane integrity and enzyme activity (i.e. cytochrome P450, isotype 3A4) over a period of almost two weeks when contained in a spheroid liver microtissue. This allows for drug toxicity testing over the same period.