Research efforts increasingly incorporate cell-based assays to obtain greater physiological relevance in disease models. Concurrently, researchers are charged with gathering and analyzing data in shorter timeframes and with greater precision, thus setting the stage for automated workflow processes. Automated systems are available in a variety of throughputs and capabilities, but an affordable automated system capable of maintaining cell health over long periods for multiple microplates has been elusive. Most automated systems are semi-automated, where liquid handling instruments perform reagent additions; but any required incubations under CO2/O2 conditions require a manual intervention by the operator. High throughput automated systems are viable alternatives for those that can afford them, but many users don’t need high throughput and its associated expense.
 Figure 1AHere, we describe an affordable walkaway assay workstation, consisting of the BioSpa™ 8 Automated Incubator, linked with BioTek liquid handling instruments and microplate readers/imagers. BioSpa 8 uses an integrated gripper to repeatedly manipulate and store up to eight lidded or unlidded microplates or other sample vessels, with up/down movement to access multiple vertical positions in the automated incubator, and left/right motion via its linear
Figure 1AHere, we describe an affordable walkaway assay workstation, consisting of the BioSpa™ 8 Automated Incubator, linked with BioTek liquid handling instruments and microplate readers/imagers. BioSpa 8 uses an integrated gripper to repeatedly manipulate and store up to eight lidded or unlidded microplates or other sample vessels, with up/down movement to access multiple vertical positions in the automated incubator, and left/right motion via its linear 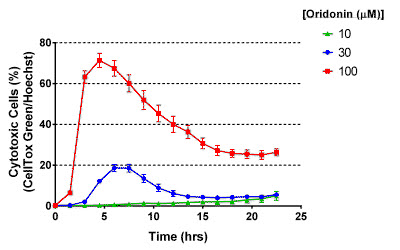 Figure 1Baxis to shuttle microplates to and from other instruments. It also maintains temperature and CO2/O2 gas levels along with humidity monitoring to maintain an ideal environment for cell cultures similar to a standard laboratory CO2 incubator. One advantage of this flexible linked system is that it provides walkaway workflow automation across a wide range of assays, including long-term live cell-based assays. It does so without the expense of a higher throughput system or the added hassle and variability due to manual intervention in the workflow. It is also useful during assay set-up for tasks such as seeding cells in microplates and performing media exchanges prior to the addition of assay reagents. Additionally, the entire automated system conveniently fits inside a conventional 6-foot biosafety cabinet for applications requiring sterility. In the following examples, we show this system applied to a long term cytotoxicity assay and a phenotype microarray, where the BioSpa 8 is interfaced to BioTek’s MultiFlo™ FX Multi-Mode Dispenser and Cytation™ 5 Cell Imaging Multi-Mode Reader and works in conjunction with their respective software programs to manage the full assay workflows without human intervention.
Figure 1Baxis to shuttle microplates to and from other instruments. It also maintains temperature and CO2/O2 gas levels along with humidity monitoring to maintain an ideal environment for cell cultures similar to a standard laboratory CO2 incubator. One advantage of this flexible linked system is that it provides walkaway workflow automation across a wide range of assays, including long-term live cell-based assays. It does so without the expense of a higher throughput system or the added hassle and variability due to manual intervention in the workflow. It is also useful during assay set-up for tasks such as seeding cells in microplates and performing media exchanges prior to the addition of assay reagents. Additionally, the entire automated system conveniently fits inside a conventional 6-foot biosafety cabinet for applications requiring sterility. In the following examples, we show this system applied to a long term cytotoxicity assay and a phenotype microarray, where the BioSpa 8 is interfaced to BioTek’s MultiFlo™ FX Multi-Mode Dispenser and Cytation™ 5 Cell Imaging Multi-Mode Reader and works in conjunction with their respective software programs to manage the full assay workflows without human intervention.
Cytotoxicity automated workflow
 Figure 2: Biolog phenotype data
Figure 2: Biolog phenotype data
Using the automated system in a biosafety hood, U-2 OS cells were dispensed into four separate microplates, which were then incubated overnight with real-time control and monitoring of environmental conditions (37 ºC, 5% CO2) and humidity levels. A series of staggered dispense routines were pre-programmed such that various concentrations of the diterpenoid, oridonin, with known anti-tumor properties, were added to a different microplate column every two hours, maintaining the environmental conditions between reagent additions. Ultimately, the microplate rows contained decreasing oridonin concentrations and the columns represented decreasing total incubation times. Software session timelines and environmental reports allow quick scrutiny of the process and system status, sending text or email notifications and alerts to bypass the need for onsite monitoring. Cells were treated with various concentrations of oridonin along with Hoechst 3342 and Promega’s CellTox™ Green to stain total and cytotoxic cells, respectively, and the plates were automatically imaged every 1.5 hours using Cytation 5’s DAPI and GFP channels. Oridonin’s cytotoxicity effects on the cells, per Figure 1, are consistent with expectations, and automated scheduling of sample preparation and data collection offers improved consistency and unattended operation versus manual methods.
Phenotype microarray automated workflow
To further illustrate efficient automation with minimal manual intervention, multiple metabolic pathways were tested simultaneously using Biolog’s Phenotype MicroArray™ along with the automated workflow, consisting of BioSpa 8, MultiFlo FX, and Cytation 5. Here, U-2 OS, HEK 293, NIH3T3, and MDA-MG-231 cells were resuspended in appropriate media, dispensed into Phenotype MicroArray panels M1 to M4, and incubated in the BioSpa 8 for 48hours at 37 ºC, 5% CO2. This incubation allows the cells to adapt and consume any residual nutrients prior to phenotype testing. Additionaly the long incubation window lends itself to running multiple assays, or co-incubating with a short-term assay, concurrently, to further enhance laboratory efficiency. After incubation, formazan dye was added to the wells, and Cytation 5 was used to measure kinetic absorbance at 590 nm every 20 minutes for 12 hours. A heat map showing the change in absorbance over the first two hours (Figure 2) shows that the cell lines react differently to the sampling of substrates, and this representation can be carried through to all 367 metabolic substrates tested.
Conclusion
The two assays described involved extensive cell-based assay workflows. Each of these workflows have been completely automated with cost-effective instrumentation providing both high precision results and freedom to the operator to perform other tasks. In addition, the Cytation 5 Cell Imaging MultiMode Reader is capable of using both CCD-based microscopy and PMT-based multimode detection for a wide range of cell-based assays, as demonstrated by these two examples using fluorescence microscopy and colorimetric detection.












