Gas chromatography (GC) serves as a primary analytical workhorse for this application due to its strong ability to separate and detect volatile analytes. Because trace monomer residues in plastics can migrate out of the material and into surrounding environments—such as packaged food, drinking water, or medical fluids—they pose potential toxicological risks.
As regulatory agencies tighten permissible exposure limits for compounds like styrene, vinyl chloride, and monomers such as bisphenol A (BPA), analytical teams are often challenged to balance the need for ultra-low detection limits with efficient sample throughput. Successfully analyzing trace monomer residues in plastics requires a solid understanding of sample extraction techniques, matrix interference, and instrument optimization to help protect both consumer safety and laboratory efficiency.
What are the regulatory standards for trace monomer residues in plastics?
Analytical laboratories testing plastics navigate strict global regulatory frameworks that dictate maximum migration limits for residual monomers. The US Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) enforce rigorous standards for food contact materials. Under the European Union’s Regulation (EU) No 10/2011, specific migration limits (SMLs) are established for hundreds of individual monomers and additives, often requiring detection limits in the parts-per-billion (ppb) range depending on the substance.
Similarly, manufacturers of medical devices and pharmaceutical packaging are expected to comply with ISO 10993 standards and various pharmacopeia directives. These regulations often involve extractables and leachables (E&L) studies. Compliance involves not only achieving the necessary limits of detection (LOD) but also maintaining comprehensive documentation of instrument calibration, system suitability, and method validation. Failure to detect trace monomer residues accurately can lead to costly product recalls, compromised public health, and potential loss of laboratory accreditation.
How does sample preparation impact GC analysis of trace monomer residues in plastics?
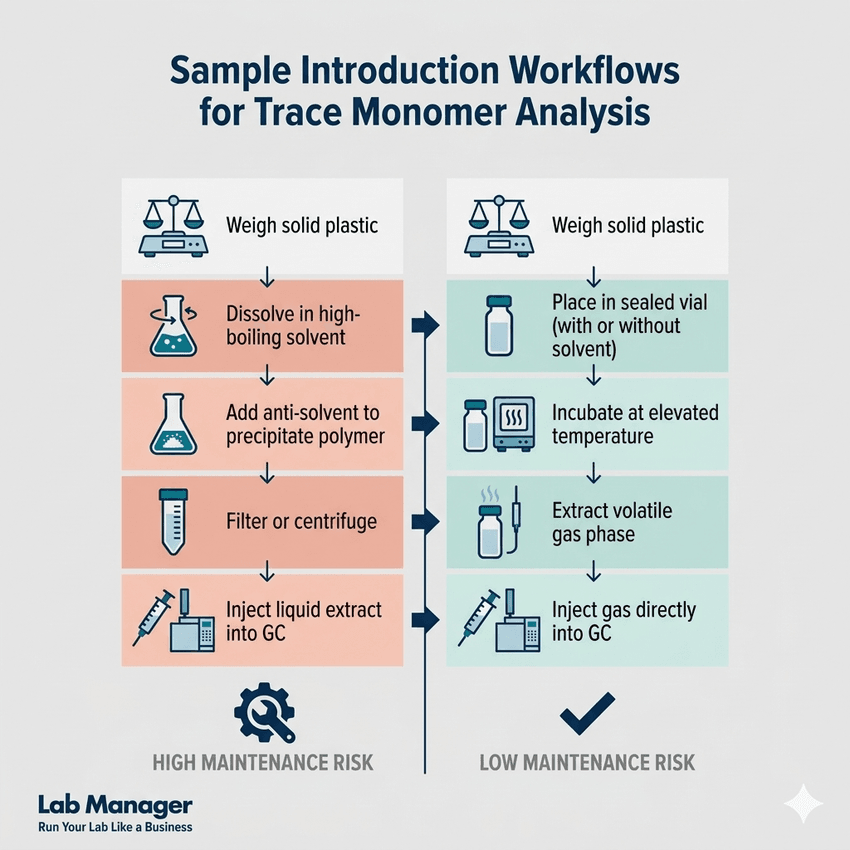
Headspace-GC vs. Liquid Injection workflows: Streamlining trace monomer analysis to reduce instrument maintenance.
GEMINI (2026)
The accuracy of a GC method is fundamentally influenced by the quality of sample preparation. Because plastics are solid, complex matrices, they cannot be injected directly into a gas chromatograph. The target monomers typically need to be extracted from the polymer network. Analysts must then transfer them into a liquid or gas phase suitable for instrument introduction. This extraction phase is frequently the most labor-intensive step in the analytical workflow.
Traditional sample preparation involves dissolution and precipitation. First, the plastic sample is dissolved in a high-boiling-point solvent, such as dimethylacetamide (DMAc) or dimethylformamide (DMF). Next, an anti-solvent is added to precipitate the high-molecular-weight polymer. This leaves the volatile monomers trapped in the liquid phase. The remaining liquid is then injected into the GC.
While effective, this method carries a high risk of introducing non-volatile polymer oligomers into the GC inlet. Such contamination can rapidly degrade column performance and necessitate frequent, costly instrument maintenance. To mitigate these risks, many high-throughput laboratories have shifted toward headspace extraction techniques.
Why is headspace gas chromatography used to analyze trace monomer residues in plastics?
Headspace gas chromatography (HS-GC) has become a highly favored technique for analyzing trace monomer residues in plastics. Instead of injecting a liquid extract, HS-GC involves placing the solid polymer (or a dissolved polymer solution) into a sealed vial. The vial is heated to a controlled temperature. This heat drives the volatile monomers out of the plastic and into the gaseous headspace above the sample.
A specialized autosampler then extracts a precise volume of this gas and injects it into the GC column. Simply put, this means the instrument only "sees" the clean gases, keeping the complex, sticky polymer matrix completely out of the system. This approach significantly reduces matrix-induced baseline drift and extends the operational lifespan of expensive consumables.
Table 1 outlines the operational differences between direct liquid injection and headspace analysis.
Table 1: Comparison of GC sample introduction techniques for plastics.
Feature | Direct Liquid Injection | Headspace Gas Chromatography (HS-GC) |
|---|---|---|
Matrix Interference | High (polymer precipitation risks) | Low (primarily volatile gases analyzed) |
Sample Prep Labor | Extensive (dissolution, filtration) | Minimal (weighing, sealing vials) |
Instrument Maintenance | High (frequent inlet and column changes) | Low (cleaner sample pathway) |
Target Analytes | Semi-volatiles and high-boiling compounds | Highly volatile and semi-volatile monomers |
Throughput | Moderate | High-throughput (easily automated) |
How can analytical teams optimize GC method parameters for trace monomer detection?
Achieving reliable trace-level quantification requires analytical teams to carefully select detectors, columns, and calibration strategies. Choosing the right equipment setup ensures the system is sensitive enough to see tiny amounts of specific chemicals.
For routine quantification of known monomers at parts-per-million levels, a Flame Ionization Detector (FID) offers a robust, cost-effective, and highly linear response for most organic compounds. However, when regulatory compliance demands ultra-trace detection (parts-per-billion or lower), or when analyzing unknown leachables, pairing the GC with a Mass Spectrometer (GC-MS) is often recommended. A mass spectrometer provides exceptional sensitivity and allows analysts to positively identify compounds based on their unique mass spectral fragmentation patterns.
Column selection is equally important. Thick-film stationary phases are generally preferred for monomer analysis, as they provide greater retention and separation of highly volatile compounds that might otherwise elute too quickly on standard columns.
Finally, managing matrix effects during calibration is central to accurate quantification. Because the release rate of a monomer into the headspace can vary depending on the density and composition of the plastic matrix, standard external calibration curves may lead to inaccurate results. Laboratories often utilize internal standards—adding a known concentration of an isotopic analog to the sample—or employ the method of standard addition to mathematically correct for matrix-induced partitioning variations.
Conclusion: Improving data integrity when evaluating trace monomer residues in plastics
Safeguarding public health and maintaining regulatory compliance relies heavily on a facility's ability to accurately detect trace monomer residues in plastics. QC personnel should carefully evaluate the capital costs of automated headspace systems and advanced detectors against the potential long-term operational savings. These savings are gained through reduced sample preparation time and lowered instrument maintenance. By refining extraction methodologies, selecting appropriate analytical columns, and implementing rigorous matrix-matched calibration strategies, analytical teams can optimize high-throughput workflows while consistently delivering legally defensible, high-quality data.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.
















