

Cross-contamination of human cell lines with other cell lines and/or misidentification of cell cultures is reported to be 18-36 percent. Find out one way to prevent contamination in your lab.


Problem: Cross-contamination of human cell lines with other cell lines and/or misidentification of cell cultures is reported to be 18−36 percent.1,2,3 Contamination can be from other human cell lines (intraspecies contaminant) or cell lines of different species altogether (interspecies contaminant). Generation of erroneous data with subsequent publication not only affects the individual laboratory utilizing the contaminated culture, it also has negative consequences for the entire research community.4 Use of contaminated cell cultures impacts research significantly both in terms of wasted financial resources and time. The continued high prevalence of cell line contamination and the consequences of utilizing these contaminated cultures highlight the need to establish the identity of cell lines being used in a research study, as well as detecting and eliminating contaminated cell cultures as quickly as possible.
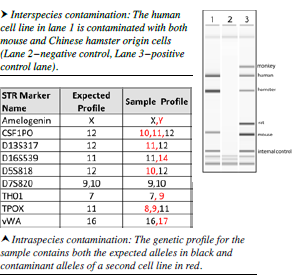
Solution: One way to deal with the cell contamination problem is through something like CellCheck,™ a comprehensive cell line authentication service that combines interspecies and intraspecies testing to verify the identity of human cell lines. Testing for interspecies contamination is performed by multiplex PCR and identifies DNA from the species of cell lines that are most commonly used in biomedical research, including: Mus musculus (laboratory mouse), Rattus norvegicus (laboratory rat), Cricetulus griseus (Chinese hamster) and Chlorocebus pygerythrus (formerly Cercopithecus aethiops[African Green Monkey]). Screening is also available for less commonly used animal cell lines, including: Sus scrofa (pig), Oryctolagus cuniculus (domesticated rabbit), Canis familiaris (dog), and Felis catus (cat).
Testing for intraspecies contamination and/or misidentification is performed by Short Tandem Repeat (STR) analysis utilizing human specific markers and verifies that the genetic profile of the sample matches the known profile of the cell line.
STR profiling is a useful quality control procedure for any human cell line or tumor. After the initial profile is determined, the cells can subsequently be retested and compared back to the original profile. STR profiling is a useful monitoring tool for tumor banks, cell culture facilities and any research study working with human cellular material. Establishing a baseline STR profile on human origin cellular research materials allows for subsequent monitoring and confirmation of the identity of the human origin materials being utilized in a research study.
 Since most cell line monitoring programs are in their infancy, guidelines on when to test and how often to monitor have yet to be established. However, the following recommendations can be used as a guideline when designing and implementing a cell line monitoring program in your laboratory: 1) authenticate cell lines at the beginning and the end of a study, 2) authenticate frozen materials stored for future use, 3) consider quarterly to semiannual testing of cell lines in continuous use, and 4) consider more frequent testing for experiments in which cell line growth is reduced (for example, cell cultures treated with therapeutic agents, cells grown in media in which factors critical for growth are removed, etc.), as these compromised cultures are more susceptible to overgrowth by contaminating cells.
Since most cell line monitoring programs are in their infancy, guidelines on when to test and how often to monitor have yet to be established. However, the following recommendations can be used as a guideline when designing and implementing a cell line monitoring program in your laboratory: 1) authenticate cell lines at the beginning and the end of a study, 2) authenticate frozen materials stored for future use, 3) consider quarterly to semiannual testing of cell lines in continuous use, and 4) consider more frequent testing for experiments in which cell line growth is reduced (for example, cell cultures treated with therapeutic agents, cells grown in media in which factors critical for growth are removed, etc.), as these compromised cultures are more susceptible to overgrowth by contaminating cells.
For more information, visit www.idexxbioresearch.com
References:
1. Lacroix M. Persistent use of “false” cell lines. Int. J. Cancer. 2008; 122:1-4.
2. Chatterjee R. Cell biology. Cases of mistaken identity. Science. 2007; 16:928-931.
3. Hughes P, Marshall D, Reid Y, Parkes H, Gelber C. The costs of using unauthenticated, over-passaged cell lines: how much more data do we need? Biotechniques. 2007; 43:575-583.
4. Torsvik A, Rosland GV, Svendsen A, et al. Spontaneous Malignant Transformation of Human Mesenchymal Stem Cells Reflects Cross-Contamination: Putting the Research Field on Track. Cancer Research. 2010; 70:6393-6396.

CURRENT ISSUE - April 2026
Why Performance, Cost, and Environmental Impact Now Compete in Procurement

