Ensuring the safety and purity of drug products requires precise residual solvent analysis during pharmaceutical manufacturing. Residual solvents—volatile impurities used during synthesis—are frequently utilized in the production of active pharmaceutical ingredients (APIs), excipients, and final drug products. As these chemicals provide no therapeutic benefit and pose toxicity risks to patients, global health authorities strictly regulate their presence in final pharmaceutical products.
For laboratory managers overseeing quality control (QC) facilities, maintaining compliance with these global standards demands robust analytical workflows. Gas chromatography (GC) is widely regarded as the gold standard for separating and quantifying these volatile compounds. However, achieving consistent, reproducible results requires a deep operational understanding of sample introduction, column chemistry, and detection limits.
Laboratories must optimize every step of the gas chromatography workflow to prevent bottlenecks and system suitability failures. Balancing high sample throughput with the acute sensitivity required to detect parts-per-million (ppm) trace impurities is a complex task. It dictates how facilities select equipment, manage consumables, and train personnel for routine testing.
What are the regulatory frameworks for residual solvents?
Pharmaceutical testing laboratories must adhere to strict, harmonized regulatory frameworks that classify solvents based on their potential risk to human health. The primary regulatory drivers for this testing are the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) and the United States Pharmacopeia (USP).
The ICH Q3C guidelines categorize residual solvents into three distinct classes based on toxicity:
- Class 1 solvents: Known human carcinogens and strongly suspected environmental hazards (e.g., benzene, carbon tetrachloride). These should be avoided entirely in pharmaceutical manufacturing unless their use is unavoidable, in which case they are strictly limited to low ppm levels (e.g., 2 ppm for benzene, per the ICH Q3C (R8) guidance).
- Class 2 solvents: Non-genotoxic animal carcinogens or possible causative agents of other irreversible toxicity (e.g., methanol, acetonitrile, dichloromethane). Their usage is limited by established Permitted Daily Exposure (PDE) limits.
- Class 3 solvents: Solvents with low toxic potential to humans (e.g., ethanol, acetone, ethyl acetate). These have a PDE of 50 mg or more per day and are generally limited to 0.5% (5000 ppm) in the final product when no specific PDE is provided.
To test for these classes, laboratories globally rely on USP General Chapter <467>, which provides standardized analytical procedures for the identification, confirmation, and quantification of residual solvents using gas chromatography. Similarly, the FDA provides specific guidance harmonized with ICH Q3C to ensure that manufacturers properly document and control volatile impurities during the drug approval process.
Why is static headspace the preferred sample introduction method?
Direct liquid injection is rarely used for residual solvent analysis in finished pharmaceuticals because APIs and excipients are typically non-volatile. Injecting these solid matrices directly into a GC system would rapidly contaminate the inlet liner, degrade the analytical column, and require extensive instrument downtime for maintenance.
To overcome this, static headspace (HS) sampling is the preferred sample introduction technique. In this method, the solid or liquid pharmaceutical sample is dissolved in a high-boiling-point diluent—most commonly water, dimethyl sulfoxide (DMSO), or dimethylformamide (DMF)—inside a precisely sealed vial. The vial is heated and agitated in an automated incubator until thermodynamic equilibrium is achieved between the liquid phase and the gas phase (the "headspace").
A precise volume of this gas phase, which contains the volatile residual solvents, is then extracted via a heated syringe or transfer line and injected into the GC. This technique isolates the volatile impurities from the heavy pharmaceutical matrix, preserving column lifespan, minimizing background noise, and enabling high-throughput automation. QC teams must carefully optimize headspace parameters, including incubation temperature, equilibration time, and vial pressurization, to ensure maximum sensitivity and reproducibility for specific solvent classes.
How do you select the right GC column and detector?
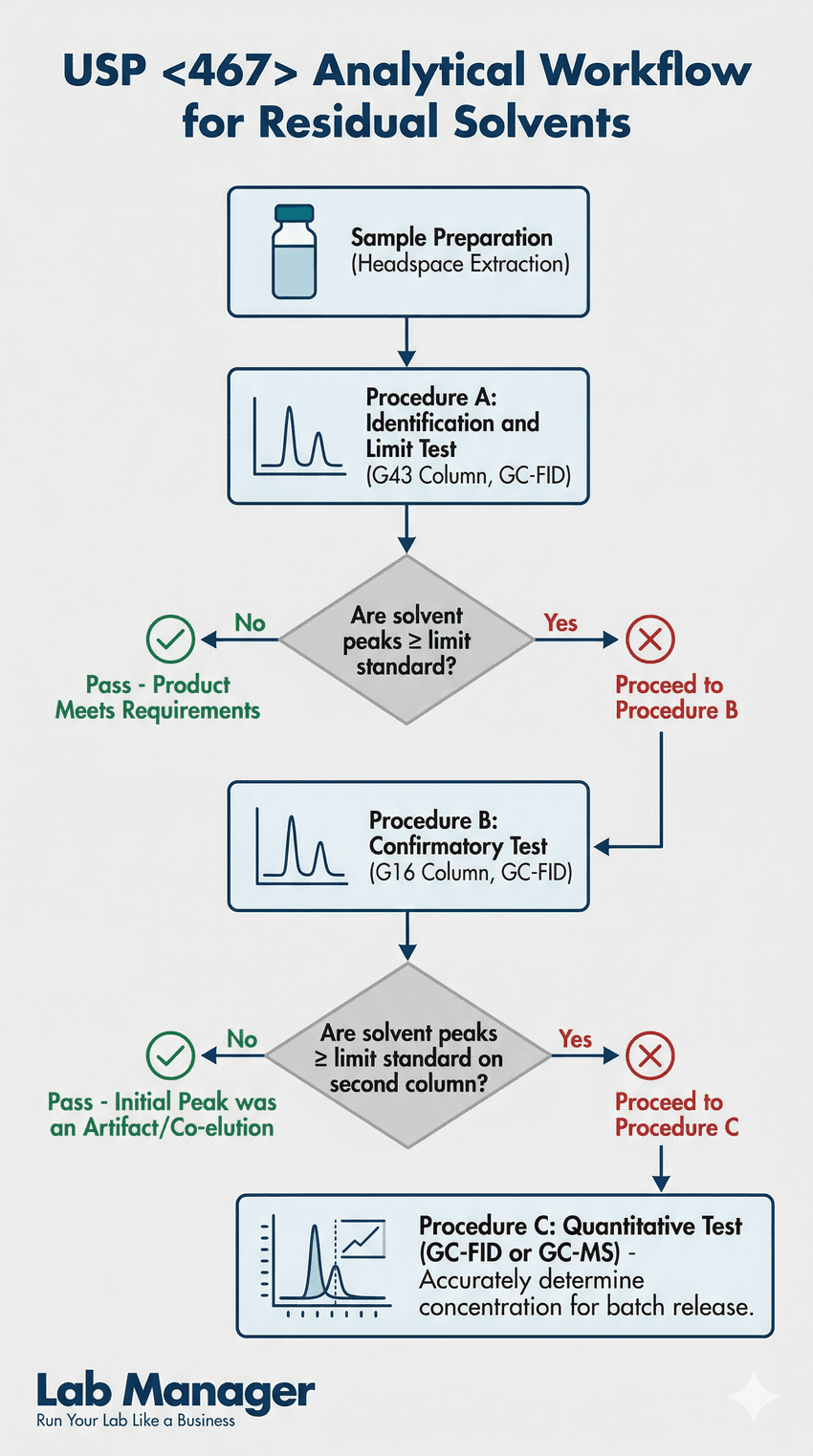
This vertical flowchart details the sequential steps to identify, confirm, and quantify residual solvents, providing a clear path from headspace extraction to final batch release.
GEMINI (2026)
The separation of complex solvent mixtures requires specific stationary phases. USP <467> outlines distinct analytical columns depending on the phase of the testing procedure.
For the initial limit test (Procedure A), the method specifies a G43 column (a mid-polarity phase consisting of 6% cyanopropylphenyl and 94% dimethyl polysiloxane). If a solvent is detected above the acceptable limit, the lab must perform a confirmation test (Procedure B) using a phase with different selectivity to prevent co-elution. This is typically done using a G16 column (a high-polarity phase comprised of polyethylene glycol). Because these columns rely on thick films (often 1.4 µm to 3.0 µm) to retain highly volatile compounds, they require optimized carrier gas flows and precise temperature programming.
Detector selection is equally critical. Flame Ionization Detectors (FID) and Mass Spectrometry (MS) are the most common choices for residual solvent analysis, as detailed in Table 1.
Table 1: Comparison of typical detectors for residual solvent analysis.
Detector Type | Sensitivity | Linear Range | Primary Application in USP <467> |
|---|---|---|---|
Flame Ionization Detector (FID) | Parts per million (ppm) | Typically wide (10^7) | Procedures A and B; routine QA/QC limit testing and quantification. Cost-effective and robust. |
Mass Spectrometer (MS) | Parts per billion (ppb) | Typically moderate (10^5) | Procedure C; definitive identification and trace quantification. Ideal for resolving co-eluting peaks or unknown impurities. |
*Note: Linear ranges are typical estimates and vary by instrument vendor and specific analytical configuration.
While FID is the workhorse of pharmaceutical QC due to its reliability and low operating cost, laboratories increasingly rely on GC-MS for complex matrices where unexpected impurities or severe co-elutions cause routine FID methods to fail.
What are common troubleshooting challenges in solvent analysis?
Even with established regulatory methods, analyzing residual solvents presents routine troubleshooting challenges that analytical teams must anticipate. Matrix effects are a primary concern; active pharmaceutical ingredients can interact with the diluent, altering the partition coefficient of the volatile solvents and causing poor recovery. Laboratories must frequently validate their methods using matrix spiking to ensure accurate quantification.
Carryover is another frequent issue, particularly when analyzing Class 2 or Class 3 solvents at high concentrations. Solvents can linger in the headspace transfer line or the syringe, contaminating subsequent blank injections. To mitigate this, facilities should ensure automated cleaning cycles are programmed into the sequence and that transfer lines are maintained at appropriately high temperatures to prevent condensation.
Finally, system suitability failures often point to column degradation. This is frequently observed as poor peak resolution between critical pairs (e.g., benzene and 1,2-dichloroethane). Thick-film GC columns are highly susceptible to oxidation. Implementing strict leak-checking protocols for carrier gases and utilizing high-capacity oxygen traps can significantly extend column life and reduce instrument downtime.
How does informatics integration ensure data integrity?
Managing the sheer volume of chromatograms and calibration data generated during routine lot-release testing requires a robust Laboratory Information Management System (LIMS) and Chromatography Data System (CDS). Regulatory agencies, particularly the FDA under 21 CFR Part 11, mandate strict data integrity controls for pharmaceutical manufacturing.
A properly integrated CDS/LIMS environment automates data transcription, minimizing human error during quantitative calculations. More importantly, it provides an unalterable audit trail. Every action—from the integration parameters applied to a specific peak to the approval of the final batch report—must be securely logged and attributable to a specific analyst. For lab managers, investing in automated, compliant data workflows drastically reduces the administrative burden of review and provides critical defense during regulatory audits.
Conclusion: Optimizing QA/QC for volatile impurities
Accurate residual solvent analysis is a non-negotiable component of pharmaceutical manufacturing that directly impacts patient safety. Quality control leaders must proactively address the operational challenges of gas chromatography. This requires standardizing robust headspace extraction protocols and selecting the optimal columns for distinct regulatory procedures. Facilities must also anticipate common troubleshooting scenarios. By aligning analytical instrumentation capabilities with strict ICH and USP requirements, and underpinning those processes with secure data management, laboratories can achieve high sample throughput. Ultimately, this ensures efficient and compliant product release.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.
















