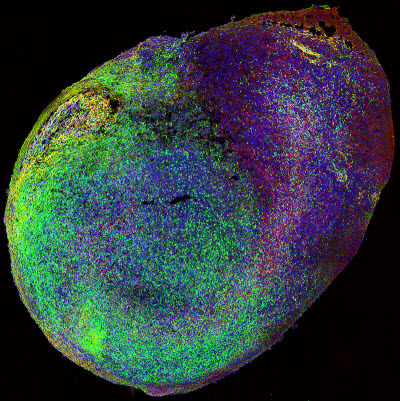
 Image courtesy of the Institute of Molecular BiologyIn 2013, researchers led by Institute of Molecular Biotechnology (IMBA) scientist Jürgen Knoblich flabbergasted the scientific community. Starting from human stem cells, researchers in his lab managed to grow living three dimensional models of basic units of human brain in a dish. These so called cerebral organoids present an unprecedented 3D cell culture model of human brain development, and have a tremendous potential for medical applications. Thanks to this discovery, scientists can examine how networks of living human brain cells develop and function, and how they are affected by different drug compounds or genetic modifications.
Image courtesy of the Institute of Molecular BiologyIn 2013, researchers led by Institute of Molecular Biotechnology (IMBA) scientist Jürgen Knoblich flabbergasted the scientific community. Starting from human stem cells, researchers in his lab managed to grow living three dimensional models of basic units of human brain in a dish. These so called cerebral organoids present an unprecedented 3D cell culture model of human brain development, and have a tremendous potential for medical applications. Thanks to this discovery, scientists can examine how networks of living human brain cells develop and function, and how they are affected by different drug compounds or genetic modifications.
For several years, Knoblich and colleagues have been working intensively to model the complex arrangement of different brain regions—a hallmark of the human brain—in order to apply this technology toward investigating even more sophisticated cell biology that could be the basis of neurological diseases.
Connecting two different brain regions in a dish
In a current publication in the journal Nature Methods, they introduce a way to systematically control the development and diversity of cerebral organoids. "In our publication, we describe a method to combine different brain tissues of choice. For this purpose, we let two different ‘building blocks’ of the forebrain, namely the dorsal and the ventral part, grow together. This so-called organoid ‘fusion’ technique allows us to observe complex interactions, such as cell migration and axon growth, between different developing brain regions," explains first author Joshua Bagley, who is a postdoctoral researcher at IMBA.
The scientists could visualize and analyse the migration of inhibitory GABAergic interneurons. These interneurons play an essential role for the correct processing of brain activity. Malfunctions of these regulatory units are associated with epilepsy, schizophrenia, and autism. For instance, inhibitory interneurons control the activity level within local brain circuits and ensure that they do not emit electrical signals excessively. A loss of this inhibition, possibly by defective cell migration, can lead to abnormal excitatory activity leading to epileptic seizures.
Wandering cells eventually get lost
In the course of brain development, interneurons are generated in a ventral part of the human brain and migrate over a long distance to their target dorsal, cortical regions, navigating themselves by chemical signals.
"It is fascinating how we can visualize interneurons wandering from the ventral to the dorsal region. These cell types orientate themselves by a process called chemotaxis, in order to find their target region. If these chemical signals are disturbed, interneurons get confused, lose track, and might not arrive at the right spot to control their neighboring neurons—epileptic seizures can be a direct consequence," illustrates Daniel Reumann, PhD student at IMBA and co-author of the study.
A protein called CXCR4 plays a key role in the migration of interneurons. The researchers deactivated this molecular signal with a drug called AMD3100, that inhibits CXCR4 and could visualize that the migration of the interneurons was impaired. This malfunction could play a role in epileptic seizures, but also potentially in other neurological disorders such as schizophrenia.
“Until a few years ago, scientists were lacking the possibilities to understanding the various causes of neurological disease sufficiently. Organoid fusions might present a paradigm shift as this new method provides us with a controlled setting to explore unsought mechanisms of epilepsy and other nervous diseases. Our technology also offers the possibility to test potential therapeutic compounds as we can use patients’ cells to grow organoids. We hope that our work will contribute to a better understanding of malfunctions that occur during the complex development of our brain and hopefully pave the way for potential treatments," states Knoblich.














