Global food safety relies heavily on analytical laboratories to rapidly identify and quantify trace pesticide residues in agricultural produce. For lab managers overseeing these high-throughput testing operations, balancing sample volume with the need for ultra-low detection limits presents a continuous operational challenge. Gas chromatography (GC), particularly when coupled with mass spectrometry, remains a foundational technology for detecting volatile and semi-volatile pesticide compounds in complex food matrices—the intricate mixture of target analytes and naturally occurring components.
As agricultural supply chains become increasingly globalized, testing facilities face mounting pressure to expand their target analyte lists while simultaneously decreasing turnaround times. Modern screening methods—such as those aligned with the European Union's SANTE guidelines for analytical quality control—reliably detect hundreds of different chemical compounds (often 200 to 500+ pesticides in a single run). These methods target varied classes, ranging from organochlorines to pyrethroids, frequently at concentrations as low as parts per billion (ppb).
Meeting these operational demands benefits from a strategic approach to instrument selection, workflow automation, and data management. Optimizing your lab's GC capabilities directly impacts the cost per sample, the validity of the analytical data, and the facility's overall reputation in a strictly regulated industry.
What are the regulatory limits for trace pesticide testing?
Food safety teams typically operate within strict regulatory frameworks that establish the Maximum Residue Limits (MRLs) for specific pesticides on agricultural commodities. In the United States, the Environmental Protection Agency (EPA) determines these legal limits, while enforcement testing is heavily guided by methodologies published in the Food and Drug Administration (FDA) Pesticide Analytical Manual. Globally, labs must also align with international standards set by bodies like the European Food Safety Authority (EFSA) and Health Canada’s Pest Management Regulatory Agency (PMRA).
Facilities are often expected to continuously validate their analytical methods to verify they meet or exceed these regulatory MRLs, which frequently vary dramatically depending on both the chemical compound and the specific crop being analyzed. Furthermore, testing facilities seeking to provide legally defensible data generally maintain ISO/IEC 17025:2017 accreditation. This involves extensive, ongoing documentation of instrument calibration, limit of quantitation (LOQ) verification, and routine proficiency testing. Failing to meet these stringent limits or maintain robust quality control can result in the release of contaminated produce or the wrongful rejection of safe agricultural products.
How does sample preparation impact GC pesticide analysis?
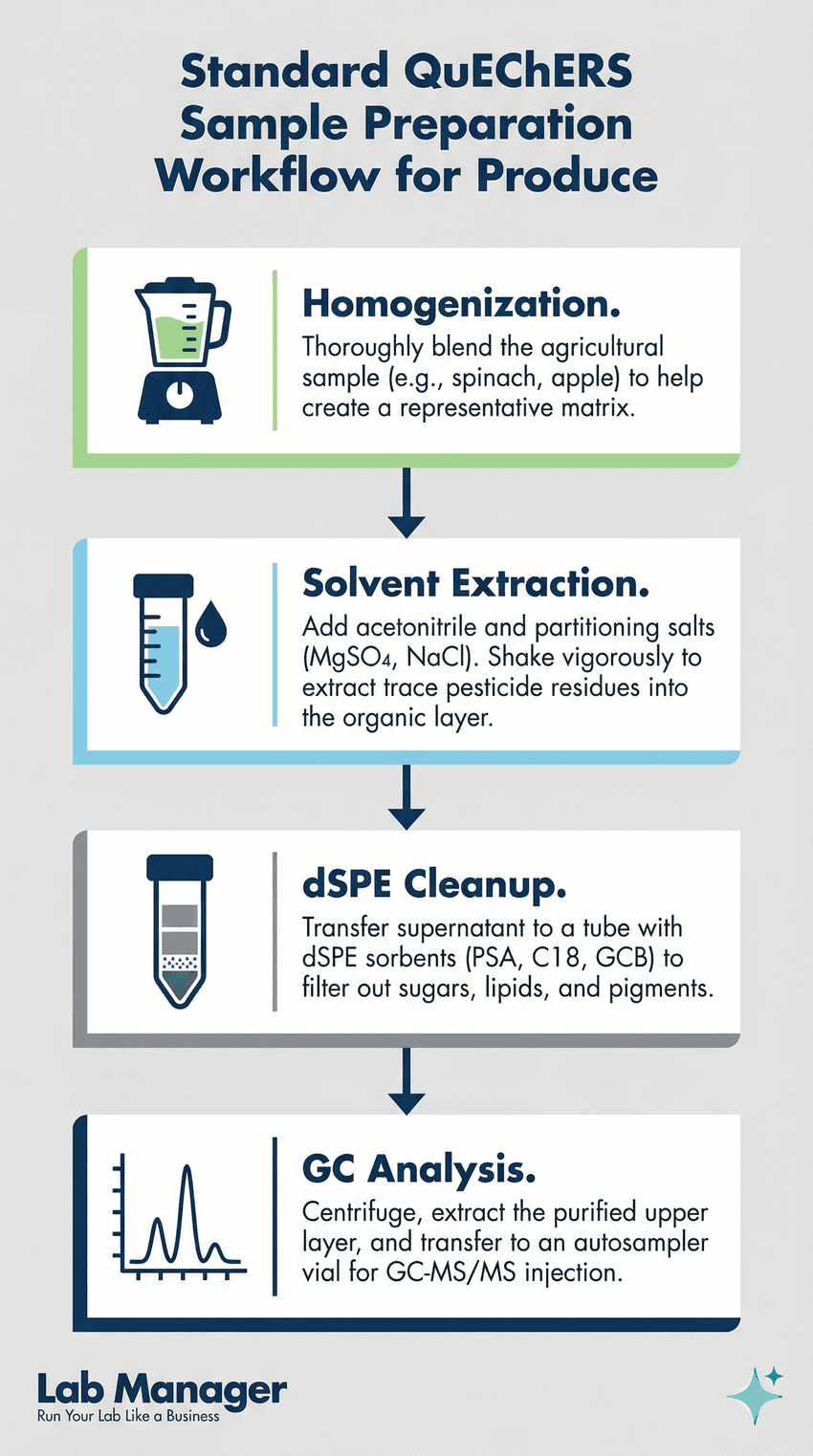
The QuEChERS method is a linear, efficient, four-step extraction and cleanup process that isolates pesticides from complex food matrices prior to analysis.
GEMINI (2026)
The accuracy and reliability of trace pesticide analysis are fundamentally influenced by the quality of the sample preparation. Agricultural produce presents a highly complex matrix filled with water, sugars, lipids, and pigments that can easily foul GC injection ports, degrade sensitive columns, and suppress analyte signals.
Historically, extracting trace pesticide residues from food matrices was a labor-intensive process requiring large volumes of hazardous solvents. Today, the industry standard has largely shifted toward the QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) methodology, recognized by the AOAC International as a highly efficient approach for multi-residue pesticide analysis.
The QuEChERS workflow typically involves two main phases:
- Extraction: The homogenized produce is shaken with a solvent (usually acetonitrile) and a mixture of partitioning salts (like magnesium sulfate and sodium chloride) to drive the pesticides into the organic solvent layer.
- Cleanup: An aliquot of the organic layer is transferred to a tube containing dispersive solid-phase extraction (dSPE) sorbents. Sorbents like PSA (primary secondary amine) remove organic acids and sugars, while GCB (graphitized carbon black) is utilized to remove pigments like chlorophyll from leafy greens.
By standardizing and streamlining this extraction process, lab managers can significantly reduce consumable costs, minimize technician exposure to toxic solvents, and vastly improve sample turnaround times. The fundamental steps of this standardized sample preparation process are detailed in Infographic 1.
How to select the right GC-MS configuration for food safety testing
When outfitting a food testing laboratory, managers frequently choose a gas chromatography configuration that balances capital expenditure with the necessary sensitivity and throughput. The choice of detector is paramount when screening for hundreds of trace pesticide residues simultaneously.
Table 1 summarizes the primary differences between the two most common GC-mass spectrometry configurations used in modern environmental and food safety labs to achieve the stringent ppb and ppt limits required by global regulatory bodies.
Table 1: Comparison of GC-MS and GC-MS/MS for pesticide analysis.
Analytical Technique | Sensitivity | Matrix Tolerance | Capital Cost | Best Application |
|---|---|---|---|---|
GC-MS (Single Quadrupole) | Parts per billion (ppb) | Low to Medium | Medium | General screening; labs testing simpler matrices or specific, targeted analyte lists. |
GC-MS/MS (Triple Quadrupole) | Parts per trillion (ppt) | High | High | Multi-residue screening; high-throughput labs analyzing complex, highly pigmented food matrices. |
While traditional single quadrupole GC-MS systems are effective for relatively clean samples, they often struggle with high background noise when analyzing complex matrices like citrus or root vegetables. Consequently, triple quadrupole GC-MS/MS systems operating in Multiple Reaction Monitoring (MRM) mode have become a standard choice for trace pesticide analysis. MRM provides exceptional selectivity by filtering out matrix ions, allowing analysts to reliably target the sub-ppb detection limits demanded by current EPA pesticide regulations.
How can labs minimize matrix effects during pesticide screening?
Even with thorough dSPE cleanup, agricultural extracts inherently contain co-extracted matrix components that enter the GC system. These components often coat the active sites within the GC inlet and analytical column. When analyzing pure solvent standards, sensitive pesticide analytes may degrade or bind to these active sites, reducing the signal. However, when these same pesticides are injected alongside a complex food matrix, the matrix components occupy the active sites instead, allowing more of the pesticide to reach the detector. This phenomenon is known as matrix-induced chromatographic response enhancement—where non-target components artificially boost the analyte's signal—a well-documented challenge in gas chromatography that has been extensively characterized by researchers such as Mastovská and Lehotay.
If a lab quantifies a produce sample against a standard prepared in pure solvent, this enhancement can lead to artificially high recovery rates and false-positive compliance failures. To overcome this, testing teams commonly utilize matrix-matched calibration. By preparing calibration standards in a "blank" matrix extract of the same crop being tested, analysts help verify that both the standards and the unknown samples experience similar enhancement effects.
Alternatively, many high-throughput facilities employ analyte protectants—compounds added to both standards and samples that intentionally coat the active sites in the GC inlet. This stabilizes the response and reduces the administrative burden of sourcing clean, pesticide-free matrix blanks for every unique crop type tested by the facility.
Why is LIMS data management critical for high-throughput pesticide testing?
Processing hundreds of multi-residue samples per week generates a massive influx of analytical data. A modern triple quadrupole GC-MS/MS method might screen for over 300 individual pesticides per injection. Reviewing this data manually is often impractical and highly prone to human error.
To manage this volume, labs frequently integrate their GC software with a robust Laboratory Information Management System (LIMS). An optimized LIMS automates the transcription of results, compares quantified levels directly against pre-programmed regulatory MRLs, and automatically flags any out-of-specification samples for review. Furthermore, LIMS platforms support complete, time-stamped traceability of the sample from receipt to final reporting, which is a key component for surviving strict regulatory audits and maintaining laboratory accreditation.
Conclusion: Optimizing your lab's GC workflow for trace pesticide analysis
Successfully testing agricultural produce for trace pesticide residues involves addressing challenges at every stage of the analytical workflow. By implementing standardized QuEChERS sample preparation, investing in highly selective GC-MS/MS instrumentation, and actively managing matrix interferences, testing facilities can reach the strict detection limits mandated by environmental and food safety regulators. Coupling these optimized physical workflows with automated LIMS data management ultimately helps labs maximize sample throughput, reduce costly rerun rates, and confidently deliver accurate, legally defensible results to the agricultural supply chain.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.
















