Contaminated glassware is one of the most preventable sources of analytical error in laboratory science. Lab glassware washers provide a validated, reproducible method for removing chemical residues, biological contaminants, and particulate matter from laboratory vessels — ensuring that each piece of equipment meets the cleanliness standard required for accurate analysis. Whether a laboratory operates under regulatory oversight or simply demands reproducible results, the performance of its glassware washing system directly influences data quality. Understanding how these instruments work, what parameters govern their effectiveness, and how to match system capabilities to application requirements is essential for any lab manager responsible for analytical accuracy.
Why glassware contamination is a critical analytical variable
Analytical errors attributed to glassware contamination are frequently underestimated. Residues from previous analyses — including traces of reagents, solvents, biological matrices, and detergent — can interfere with downstream assays, shift pH readings, introduce false positives in trace-level work, or alter chromatographic baselines. A single beaker retaining nanogram-level pesticide residue, for example, can compromise an environmental water analysis performed at parts-per-trillion detection limits.
Manual washing introduces variability that automated systems eliminate. Human scrubbing cannot guarantee consistent contact time, temperature, or mechanical action across every vessel, and manual rinsing often fails to remove all detergent traces from narrow-bore glassware or complex vessel geometries. Automated lab glassware washers address these challenges by delivering programmed wash and rinse cycles under controlled temperature, pressure, and time conditions. The result is a reproducible cleaning outcome that can be validated, documented, and audited — a critical requirement in any laboratory operating under GLP, GMP, or ISO/IEC 17025 accreditation.
The consequences of inadequate glassware cleaning extend beyond individual sample failures. In high-throughput analytical environments, systemic cleaning failures can propagate across entire analytical runs before the source is identified, resulting in costly reanalysis, delayed results, and potential regulatory findings. Building a robust glassware washing program is therefore not simply a housekeeping function — it is a foundational element of laboratory quality assurance.
How lab glassware washers work: cycles, temperature, and mechanical action
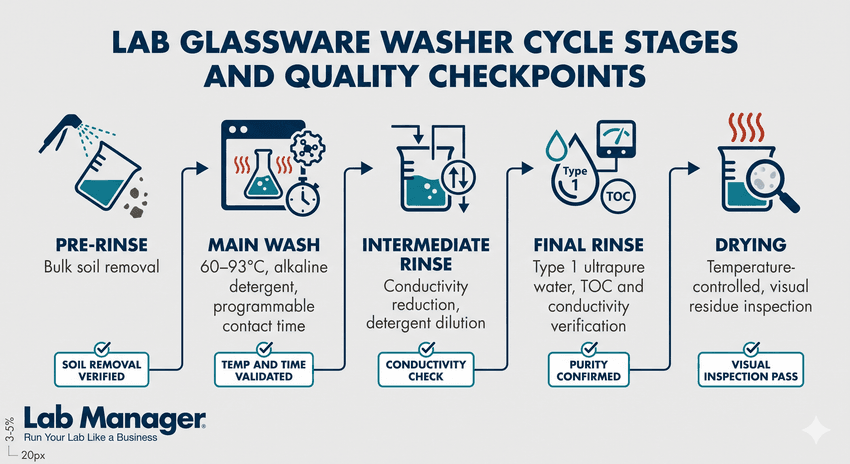
A step-by-step breakdown of a laboratory glassware washing cycle, highlighting the essential parameters and quality validation checkpoints needed at each stage to ensure properly decontaminated, residue-free equipment.
GEMINI (2026)
Laboratory glassware washers operate on a principle similar to domestic dishwashers but with significantly more precise process control. A standard wash cycle consists of several discrete phases: a pre-rinse to remove bulk contamination, a main wash with heated detergent solution, one or more intermediate rinses, a final rinse using purified water, and a drying stage. Each phase is programmable, with variables including water temperature, spray pressure, cycle duration, and detergent dosing controlled through the instrument's interface.
Temperature is among the most important parameters in the wash cycle. Most laboratory washers operate between 60°C and 93°C during the main wash phase, with higher temperatures improving both detergent activity and microbial reduction. For thermolabile residues or heat-sensitive glassware types, lower-temperature programs can be applied. Final rinse water temperature also matters: a hot final rinse improves drying efficiency and reduces waterspot formation on volumetric and optical glassware.
Mechanical action in a laboratory washer is delivered through rotating spray arms that distribute wash solution through nozzle arrays under pressure. Many instruments additionally incorporate injector systems — bottom-mounted cannulas or rack-integrated injector pins — that direct flow directly into the interior of flasks, cylinders, and tubes. This interior injection is critical for items like volumetric flasks, burettes, and condensers, where spray arm coverage alone cannot guarantee adequate internal cleaning. Balancing cleaning performance against throughput and resource consumption requires careful consideration of cycle design — particularly in high-volume labs where [managing water use and cycle efficiency in glassware washing] has a direct impact on both operational costs and turnaround times.
Detergent selection and rinse water quality
Detergent chemistry is one of the most consequential — and most frequently overlooked — variables in laboratory glassware washing. Laboratory-grade detergents are formulated to be low-foaming, compatible with automated spray systems, and residue-free after adequate rinsing. Most are alkaline, with pH values ranging from 9 to 12, making them effective at saponifying lipids, solubilizing protein residues, and dispersing inorganic particulates. Neutral and mildly acidic formulations are also available for applications where high pH is incompatible with the analysis — for instance, when washing glassware used in pH-sensitive biochemical assays or ion-selective electrode work.
Detergent concentration must be precisely controlled. Too little detergent results in inadequate soil removal; too much creates rinsing challenges and elevates the risk of surfactant carryover into subsequent analyses. Most modern lab washers include integrated dosing systems that dispense a metered volume per cycle, and regular calibration of these systems is essential — dosing drift can introduce systematic cleaning failures that may not be immediately apparent from visual inspection alone.
The quality of water used in the final rinse stage is equally critical. Tap water introduces mineral ions, chlorine, and organic contaminants that deposit on glassware surfaces after drying. Deionized water removes ionic species but may still contain residual organic impurities. Type 1 ultrapure water — produced by reverse osmosis combined with ion exchange and UV oxidation — is the appropriate final rinse medium for trace-level analytical work. Laboratories performing ICP-MS, ion chromatography, or ultra-trace environmental analysis should use only Type 1 water in the final rinse to avoid metal ion deposition or elevated organic baselines. The downstream analytical consequences of [detergent residue on glassware surfaces] are significant enough that many regulated methods require glassware blank testing as a routine quality control step.
| Rinse water type | Typical resistivity | Appropriate applications |
|---|---|---|
| Deionized (Type 3) | ≥0.05 MΩ·cm | General laboratory glassware, non-critical assays |
| Purified (Type 2) | ≥1 MΩ·cm | HPLC preparation, cell culture, routine analytical work |
| Ultrapure (Type 1) | ≥18 MΩ·cm | ICP-MS, trace metals, ultra-trace organics, pharmacopoeial testing |
Matching washer configuration to laboratory application
Laboratory glassware washers are available in several form factors, each suited to different throughput levels and installation constraints. Undercounter models are well-suited to smaller labs with moderate daily volumes and limited floor space. Freestanding and pass-through configurations offer higher capacity and are appropriate for centralized glassware processing in large analytical facilities. Some manufacturers supply modular rack systems that accommodate specialized labware — including round-bottom flasks, serological pipettes, microplates, and Kjeldahl tubes — requiring careful assessment of rack and injector compatibility before a system is specified.
Application context is the primary driver of washer selection. Pharmaceutical quality laboratories operating under FDA 21 CFR Part 211 or pharmacopoeial cleaning guidance must demonstrate that their washing process consistently delivers glassware free of detergent residue, microbial contamination, and chemical carryover. Doing so requires documented process validation, defined acceptance criteria, and — for 21 CFR Part 11 compliance — washers with data logging or electronic audit trail capability. The full requirements involved in [cleaning validation for glassware washers in pharmaceutical manufacturing environments] represent a level of process control that extends well beyond standard laboratory practice.
Food safety and beverage testing laboratories face distinct contamination challenges. Lipid residues from fatty matrices — oils, emulsified dairy products, meat extracts — are among the most tenacious foulants in analytical labware and require elevated wash temperatures, specialized alkaline detergent formulations, and extended cycle times for reliable removal. Glassware used for pesticide residue analysis must also be free of any trace organochlorine or organophosphate carryover, requiring dedicated wash programs and, in some cases, solvent pre-rinse steps. The complexities of [cleaning glassware contaminated with lipids and food matrix residues] make detergent and program selection particularly critical in food testing contexts.
Environmental laboratories performing metals analysis by ICP-OES, ICP-MS, or graphite furnace AAS face the most stringent cleanliness demands of any laboratory type. Glassware used in these applications must be free of metal ion deposits at the sub-microgram-per-liter level, typically requiring dedicated acid-washing protocols with dilute nitric or hydrochloric acid and Type 1 ultrapure final rinses. Standard alkaline wash programs that are entirely adequate for other applications are insufficient for [preparing trace-metal-free glassware for environmental water analysis]. Dedicated storage protocols — sealed containers in a clean, acid-washed environment — are also required to maintain glassware cleanliness between use.
Materials science, polymer research, and petrochemical analysis laboratories present cross-contamination risks that differ substantially from biological or environmental contexts. Hydrocarbon matrices, organic solvent residues, polymer additives, and surfactant formulations can persist through standard aqueous washing cycles. In GC and GC-MS applications, even trace-level [hydrocarbon and solvent residue carryover in petrochemical glassware] can generate ghost peaks, elevate baselines, or mask low-concentration analytes in subsequent runs. These laboratories often require specialized solvent pre-wash steps or dedicated wash programs as part of a validated cleaning workflow.
Safety considerations in automated glassware washing
Automated glassware washers reduce operator exposure to chemically contaminated labware by minimizing direct handling. However, the instruments themselves introduce safety hazards that require active management. High-temperature wash cycles present burn risks during loading and unloading, and steam released when opening the washer door immediately after cycle completion can cause injury if personnel have not been trained to allow adequate cool-down time.
Detergent handling is a second exposure route. Concentrated laboratory detergents are typically corrosive, with pH values between 11 and 13, and can cause skin and mucosal injury on contact. Personal protective equipment — chemical-resistant gloves, splash goggles, and a lab coat — must be worn during detergent cartridge replacement and system maintenance, even when integrated dosing systems are in place. The broader picture of [chemical exposure risks in automated glassware washing operations] includes not only detergent contact but also volatile residue release during hot cycle operation and aerosol generation during loading of heavily contaminated ware. All of these hazards warrant formal risk assessment and documented staff training.
Glassware breakage inside the washer is an underappreciated risk. Thin-walled volumetric ware, chipped items, or glassware improperly seated on racks can fracture under high-pressure spray or thermal shock. Labs should implement routine condition-checking protocols before loading, and racks should be configured to avoid direct nozzle impingement on fragile vessel walls. Broken glass inside the washer chamber must be removed carefully before the next cycle to prevent injury during subsequent loading.
Key safety practices for laboratory glassware washer operation:
- Allow the washer to complete its full cool-down phase before opening the door
- Wear chemical-resistant gloves and eye protection during detergent handling and maintenance
- Inspect glassware for chips, cracks, or damage before loading each cycle
- Use rack accessories rated for the vessel types being washed
- Follow the manufacturer's maintenance schedule for spray arms, door seals, and filters
- Store concentrated detergent in a locked, ventilated chemical storage cabinet
Validation and documentation requirements for glassware washing
Process validation is a regulatory requirement in GMP and GLP-regulated laboratories and a recognized best practice in any accredited analytical environment. Validation establishes that a defined wash program reliably achieves the required level of cleanliness across the full range of glassware types and soil loads encountered in routine use. It typically involves rinse-water testing — measuring conductivity, pH, and total organic carbon (TOC) in the final rinse effluent — combined with defined visual inspection criteria and, where necessary, specific chemical residue testing.
Ongoing performance monitoring should include periodic verification of detergent dosing accuracy, final rinse water quality, spray arm function, and wash temperature profiles across the full cycle. Many modern laboratory washers include onboard data logging that records cycle parameters for every run, providing a retrievable quality record. In regulated environments, this data must be retained in accordance with applicable GMP or GLP documentation requirements.
Key parameters to monitor in a glassware washer performance verification program:
- Final rinse water conductivity (target: ≤0.056 µS/cm for ASTM Type I ultrapure water; ≤1.0 µS/cm for Type II purified water rinse)
- Main wash temperature (verified against the validated range for each program)
- Detergent dose volume (verified against calibration target at defined intervals)
- Spray arm rotation and nozzle condition (visual check during scheduled maintenance)
- TOC of final rinse effluent (in trace-organic-sensitive applications)
- Post-drying visual inspection (absence of residue, water beading, or etching)
Revalidation is required following any change to wash programs, detergent supplier or formulation, washer servicing or component replacement, or changes to the types of glassware processed. All validation activities and ongoing monitoring results should be documented in a manner consistent with the laboratory's quality management system, whether that is ISO/IEC 17025, ICH Q7 (GMP for active pharmaceutical ingredients), or facility-specific SOPs.
Achieving analytical purity starts with glassware washing
Lab glassware washers are not peripheral equipment — they are a foundational element of the analytical workflow, and their performance directly determines whether downstream results can be trusted. Selecting the right washer format for the lab's throughput and space requirements, programming appropriate wash cycles for each application type, using compatible detergent chemistry, and validating the process against meaningful acceptance criteria are all essential steps in building a glassware management program that reliably supports analytical purity. As detection limits decrease and regulatory expectations intensify across analytical disciplines, the margin for glassware-related contamination errors continues to shrink. A well-specified, properly validated lab glassware washer is one of the most reliable investments a laboratory can make in the long-term integrity of its data. For labs at the point of evaluating or upgrading their systems, understanding [what to prioritize when selecting a laboratory glassware washer] is the logical first step toward a system that fits both the application and the budget.
References
- United States Pharmacopeia. USP General Chapter <1231> Water for Pharmaceutical Purposes. USP–NF. Rockville, MD: United States Pharmacopeial Convention. https://doi.org/10.31003/USPNF_M99956_07_01
- ASTM International. ASTM D1193-06(2018): Standard Specification for Reagent Water. West Conshohocken, PA: ASTM International. https://store.astm.org/d1193-06r18.html
- Prichard, E., & Barwick, V. (2007). Quality Assurance in Analytical Chemistry. Chichester: Wiley-Interscience. https://books.google.com/books/about/Quality_Assurance_in_Analytical_Chemistr.html?id=g-zH-odv29EC
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.















