Agricultural and environmental laboratories face increasing demands for high-throughput, highly accurate testing of soil and plant nutrient profiles. Inductively coupled plasma mass spectrometry (ICP-MS) has emerged as the premier analytical technique to meet these requirements, offering simultaneous multi-element analysis with exceptional detection limits. For lab managers, transitioning to or optimizing an ICP-MS workflow means balancing initial capital investment with long-term gains in sample turnaround time and data quality.
Unlike traditional atomic absorption spectroscopy, ICP-MS allows laboratories to quantify both essential macronutrients and trace heavy metal contaminants in a single analytical run. This capability is critical for agricultural operations that rely on comprehensive soil and plant nutrient data to optimize crop yields, monitor soil health, and ensure regulatory compliance regarding heavy metal uptake in food crops.
Successfully operating an ICP-MS system for agricultural samples requires more than just installing the instrument. Laboratory leaders must implement robust standard operating procedures (SOPs) for sample digestion, matrix interference mitigation, and routine instrument maintenance to ensure consistent, reliable analytical results across highly variable sample types.
How to overcome matrix challenges in soil and plant sample preparation
The accuracy of any ICP-MS analysis is fundamentally dependent on the quality of the sample preparation. Soil and plant tissues present highly complex, variable matrices that must be completely broken down into a stable, dissolved state before introduction to the plasma. For modern analytical laboratories, closed-vessel microwave digestion is the industry standard, offering superior recovery rates and reduced contamination risks compared to traditional hot block methods.
Soil samples are notoriously difficult to digest due to their high silica content and diverse mineralogy. While nitric acid and hydrochloric acid are standard for extracting trace metals and available nutrients, hydrofluoric acid is often required for total elemental recovery, which introduces significant safety and instrument compatibility concerns. Plant nutrient analysis, conversely, deals with high levels of organic matter. Incomplete digestion of plant tissue leaves residual carbon in the sample, which can severely impact plasma stability and create downstream interferences in the mass spectrometer.
Laboratory managers must standardize acid mixtures and microwave heating profiles based on the specific analytical goals of their client base. A summary of standard approaches is provided in Table 1.
Table 1: Common Digestion Reagents for Agricultural Samples
Sample Matrix | Target Analytes | Primary Acid Mixture | Key Operational Considerations |
|---|---|---|---|
Soil | Extractable metals, macronutrients | Nitric acid (HNO3) + Hydrochloric acid (HCl) | Standard EPA Method 3051A approach; leaves silicates intact. |
Soil | Total elemental recovery | HNO3 + HCl + Hydrofluoric acid (HF) | Requires specialized HF-resistant sample introduction components. |
Plant Tissue | Micronutrients, heavy metals | HNO3 + Hydrogen peroxide (H2O2) | H2O2 aggressively oxidizes organic carbon to prevent plasma loading. |
How to manage spectral and non-spectral interferences in ICP-MS
Even with perfect sample preparation, soil and plant nutrient analysis via ICP-MS is subject to significant interferences that can skew quantitative results. Non-spectral interferences, or matrix effects, occur when high concentrations of easily ionized elements (like sodium or potassium, common in agricultural soils) suppress or enhance the ionization of target analytes. To compensate, laboratories must utilize internal standards—elements like rhodium, yttrium, or scandium that are not naturally present in the samples—added online during the analytical run to correct for physical and mass-charge variations.
Spectral interferences present a more complex challenge. These occur when polyatomic ions generated in the argon plasma share the same mass-to-charge ratio (m/z) as the target analyte. For example, residual carbon from plant tissue can combine with argon to form ArC+, which interferes directly with the measurement of chromium at m/z 52. Similarly, chloride from soil extractions can form ArCl+, interfering with arsenic at m/z 75.
Modern ICP-MS systems combat these overlaps using collision/reaction cells (CRC). By introducing a gas like helium or hydrogen into the cell, polyatomic interferences are either neutralized via kinetic energy discrimination (KED) or converted into harmless byproducts through predictable chemical reactions. Lab managers should ensure their method SOPs clearly define CRC gas flows and tuning parameters to eliminate false positives in critical agricultural nutrient data.
How to optimize high-throughput ICP-MS workflows for agricultural labs
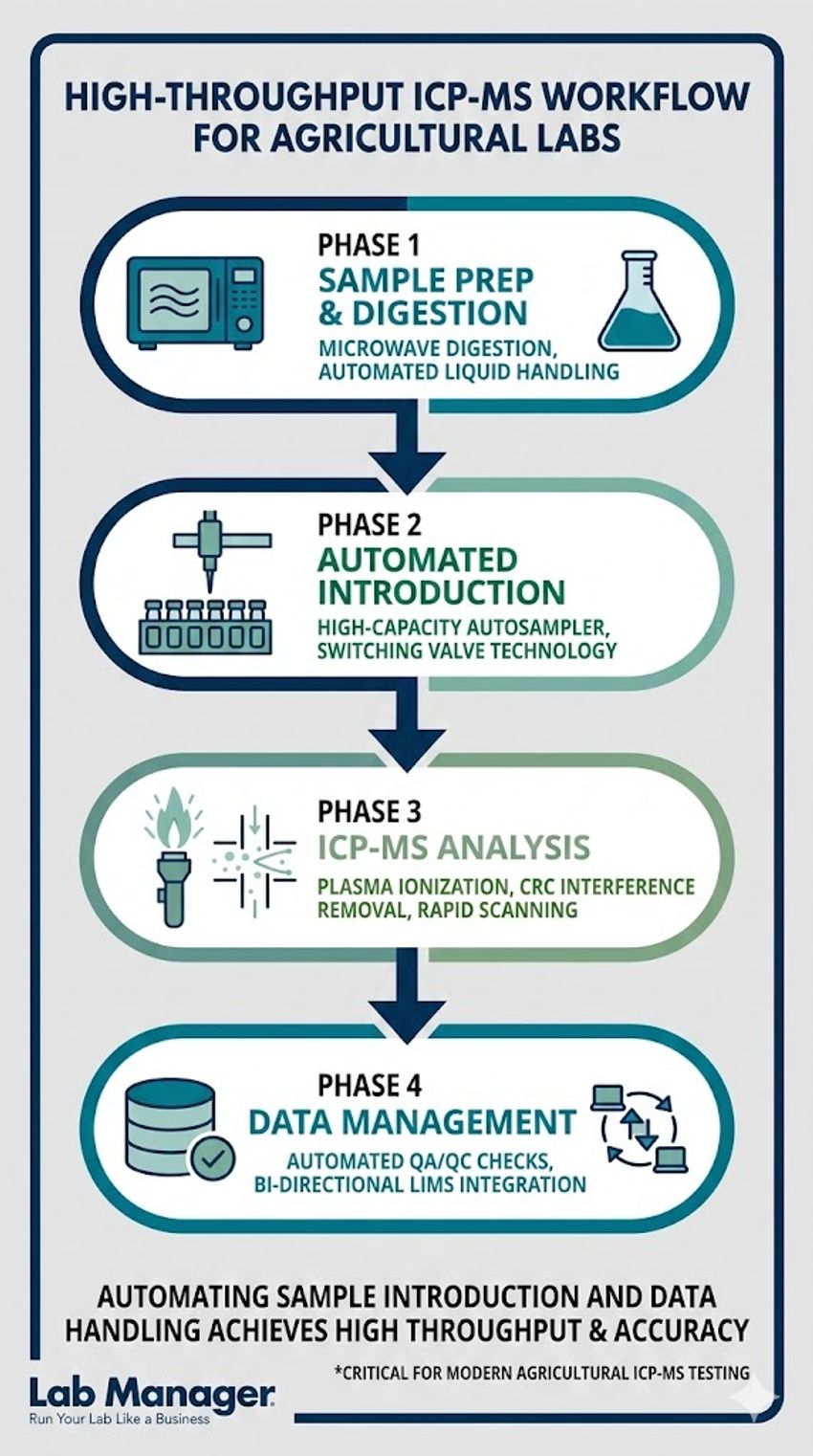
This vertical workflow illustrates the four critical phases of a high-throughput ICP-MS setup.
GEMINI (2026)
Agricultural laboratories often face seasonal surges in sample volume, particularly during pre-planting and post-harvest periods. To maintain profitability and meet tight turnaround times, lab managers must optimize the entire ICP-MS workflow for efficiency. Automation is the primary driver of this optimization.
High-capacity autosamplers paired with automated discrete sample introduction systems (often utilizing switching valves) drastically reduce sample uptake and washout times. By minimizing the time the sample spends in the introduction system, laboratories not only increase sample throughput but also reduce the wear on instrument components caused by harsh, high-matrix soil digests.
Furthermore, integrating the ICP-MS software directly with the laboratory information management system (LIMS) is essential. Bi-directional LIMS integration allows for the automated downloading of worklists and the seamless upload of finalized nutrient data, eliminating manual transcription errors and accelerating the reporting pipeline.
How to maintain ICP-MS hardware for high-matrix agricultural samples
Routine maintenance is the most critical operational variable separating highly efficient ICP-MS labs from those plagued by downtime. Soil and plant digests are characterized by high total dissolved solids (TDS). Even when diluted to the industry standard of <0.2% TDS, these samples will inevitably cause salt deposition on the instrument's interface cones (sampler and skimmer) and injector tube.
If left unchecked, cone blockage leads to signal drift, loss of sensitivity, and failed quality control checks. Lab managers must establish aggressive, strictly documented preventative maintenance schedules. Daily tasks should include inspecting and cleaning the nebulizer, spray chamber, and interface cones.
Additionally, maintaining proper exhaust ventilation and monitoring the condition of the roughing pump oil are vital for plasma stability and vacuum efficiency. By training bench personnel to proactively address hardware wear rather than reacting to failing QC data, laboratories can maximize instrument uptime during peak agricultural testing seasons.
Conclusion: Maximizing efficiency in agricultural ICP-MS testing
Implementing and managing an ICP-MS workflow for soil and plant nutrient analysis represents a significant advancement in analytical capability. By investing in robust sample digestion protocols, utilizing collision/reaction cells to mitigate interferences, and automating sample introduction, laboratory leaders can confidently deliver the high-quality, comprehensive elemental data that modern agriculture demands. Focusing on rigorous preventative maintenance and LIMS integration ensures that these high-tech systems remain reliable, profitable assets year-round.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.














