For environmental testing facilities and municipal water authorities, selecting the right analytical technology is central to laboratory operations. Using inductively coupled plasma mass spectrometry (ICP-MS) for drinking water analysis has become the industry standard for detecting trace metals and metalloids. Public health relies heavily on the ability of laboratories to accurately quantify contaminants at extremely low concentrations, often in the parts-per-trillion (ppt) range.
As regulatory limits for toxic elements like lead, arsenic, and cadmium continue to tighten globally, lab managers face the dual challenge of meeting strict compliance standards while maintaining high sample throughput. Older analytical techniques, while still useful for certain applications, often lack the sensitivity or multi-element capabilities required for modern drinking water screening.
This guide explores the operational workflows, regulatory methodologies, and technical strategies necessary to optimize ICP-MS performance. By standardizing sample preparation, managing polyatomic interferences, and automating daily tuning routines, laboratory leaders can ensure accurate, defensible data that protects public health.
Why is ICP-MS replacing older techniques in water testing?
Historically, laboratories relied on a combination of flame atomic absorption spectroscopy (FAAS), graphite furnace atomic absorption spectroscopy (GFAAS), and inductively coupled plasma optical emission spectroscopy (ICP-OES) to analyze water samples. While these methods are effective, they often require multiple analytical runs on different instruments to cover the full spectrum of regulated elements, from major minerals like calcium and magnesium to trace toxins like mercury and lead.
ICP-MS consolidates these analytical workflows. The technology uses an argon plasma operating at temperatures near 10,000 degrees Celsius to completely atomize and ionize the elements present in a liquid sample. These positively charged ions are then extracted into a vacuum system and separated by a mass spectrometer based on their unique mass-to-charge ratios.
The primary operational advantage of this technology is its exceptionally wide dynamic range. Modern mass spectrometers can measure trace contaminants at sub-ppt levels and major cations at parts-per-million (ppm) levels within a single analytical sequence. For high-volume environmental laboratories, this consolidation significantly reduces sample handling time, limits consumable usage, and lowers the overall cost per analysis.
What are the regulatory requirements for ICP-MS drinking water analysis?
Drinking water analysis is heavily governed by standardized methods that dictate instrument performance criteria, quality control guidelines, and required reporting limits. In the United States, the Environmental Protection Agency (EPA) establishes maximum contaminant levels (MCLs) for heavy metals to ensure public safety.
Analyzing drinking water for these contaminants typically follows EPA Method 200.8, which outlines the specific protocols for determining trace elements in ground, surface, and drinking waters by ICP-MS. The method defines everything from acceptable instrument tuning parameters to the required frequencies for quality control checks. Similarly, Standard Method 3125 is widely utilized by municipalities and researchers to quantify heavy metal release, such as tracking the seasonal mobilization of lead and aluminum in premise plumbing and distribution systems. Internationally, ISO 17294-2 provides the framework for water quality testing, specifying the application of ICP-MS for determining over 60 elements.
Adhering to these frameworks requires rigorous quality control procedures. Laboratories must run calibration blanks, laboratory fortified blanks (spikes), and continuing calibration verification (CCV) standards at defined intervals—usually after every ten client samples. When developing or updating standard operating procedures (SOPs), lab managers must ensure that the laboratory information management system (LIMS) is configured to automatically flag any QC recoveries that fall outside the typical 85 to 115 percent acceptance window.
How should laboratories prepare drinking water samples for ICP-MS?
The accuracy of any trace elemental analysis is highly dependent on sample preparation. Because mass spectrometry is sensitive to the parts-per-trillion level, background contamination from airborne dust, improperly cleaned labware, or impure reagents can easily compromise results.
Water samples are generally categorized into two testing requirements: dissolved metals and total recoverable metals. To measure dissolved metals, the water sample must be filtered through a 0.45-micron membrane immediately after collection to remove particulate matter. Following filtration, the sample is acidified to a pH of less than 2.0.
For total recoverable metals, the sample is not filtered. Instead, it undergoes an acid digestion process using nitric acid, and frequently, a small amount of hydrochloric acid. The addition of hydrochloric acid is particularly important for maintaining the chemical stability of elements like antimony, silver, and mercury in solution. Recent studies analyzing heavy metals in drinking water emphasize the importance of thorough digestion and acidification to ensure that elements bound to suspended particles are fully mobilized into solution before entering the instrument.
Laboratory managers should enforce strict environmental controls in the sample preparation area. Utilizing cleanroom techniques, dedicated high-density polyethylene (HDPE) or fluoropolymer vessels, and high-purity deionized water (18.2 MΩ·cm) are non-negotiable operational standards for trace-level analytical success.
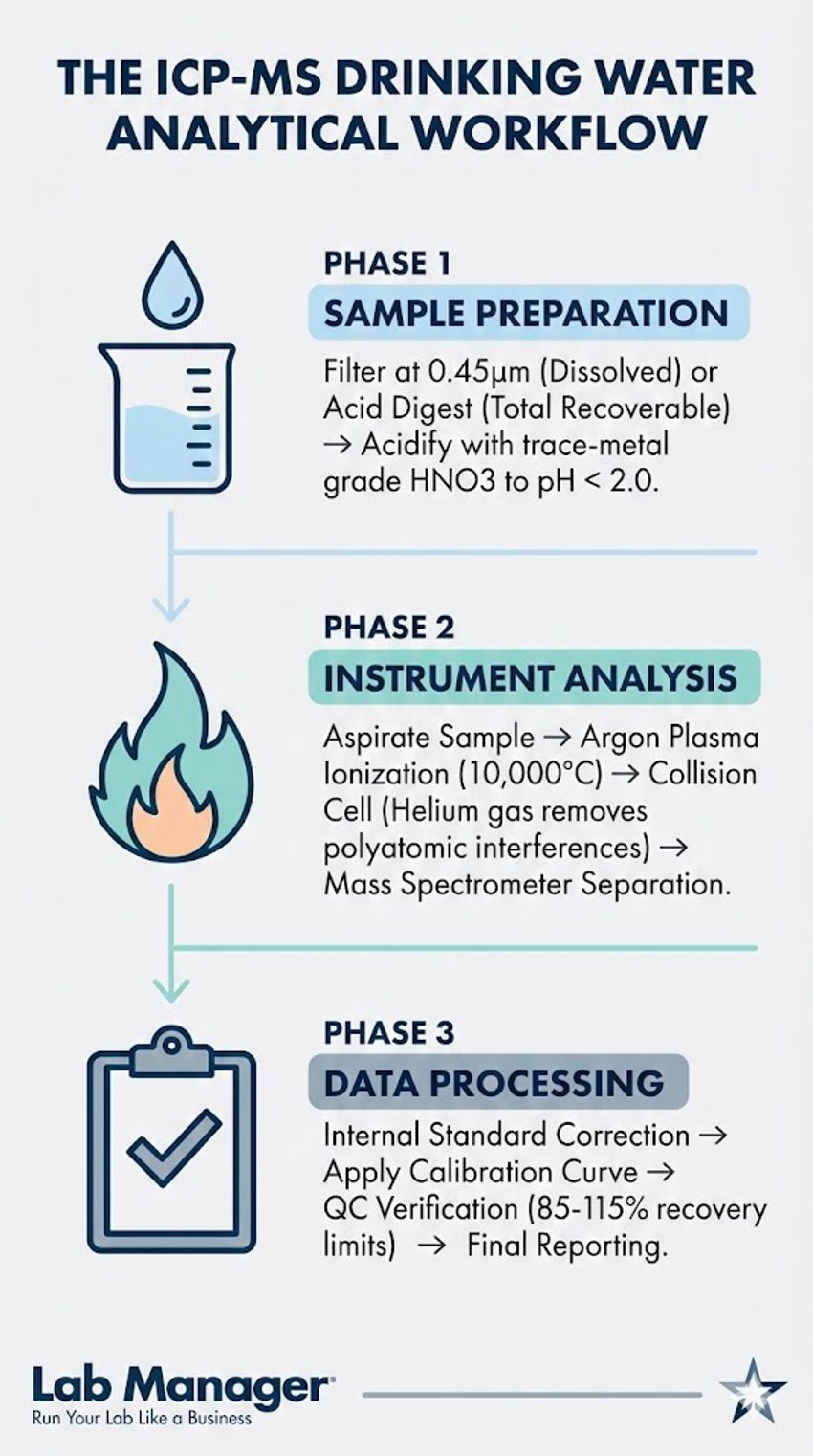
Navigating the complexities of trace elemental analysis requires a precise, phased approach.
GEMINI (2026)
How do collision cells manage polyatomic interferences in ICP-MS?
One of the primary technical challenges in ICP-MS analysis is the formation of polyatomic interferences. These occur when ions from the plasma gas, sample matrix, or surrounding atmosphere combine to form molecules with the same mass-to-charge ratio as the target analyte.
For example, in a sample containing high levels of chloride, argon from the plasma can combine with chloride to form the polyatomic ion 40Ar35Cl. This molecule has a mass of 75, which directly overlaps with the single stable isotope of arsenic (75As), leading to a false positive or an artificially elevated arsenic reading. Similarly, an argon dimer (40Ar40Ar) overlaps with the primary isotope of selenium (80Se).
Modern instruments mitigate these interferences using collision/reaction cells (CRCs). Before the ion beam enters the mass analyzer, it passes through a cell filled with a gas. Table 1 summarizes common polyatomic interferences and their corresponding collision gas solutions.
Table 1: Common Polyatomic Interferences in Drinking Water Analysis
Target Analyte | Interfering Molecule | Mass | Collision/Reaction Strategy |
|---|---|---|---|
Arsenic (As) | 40Ar35Cl+ | 75 | Helium (Kinetic Energy Discrimination) |
Selenium (Se) | 40Ar40Ar+ | 80 | Hydrogen gas reaction or Helium KED |
Chromium (Cr) | 40Ar12C+ | 52 | Helium (Kinetic Energy Discrimination) |
Vanadium (V) | 35Cl16O+ | 51 | Helium (Kinetic Energy Discrimination) |
The most common operational approach for drinking water is Kinetic Energy Discrimination (KED) using helium gas. Because polyatomic molecules are physically larger than single analyte ions of the same mass, they collide more frequently with the helium gas inside the cell. These collisions cause the polyatomic molecules to lose kinetic energy, which prevents them from entering the mass analyzer. Meanwhile, the smaller analyte ions retain enough energy to pass through successfully and be quantified.
How can labs optimize daily ICP-MS workflows and instrument tuning?
Consistent daily workflows are critical for reliable data output and minimizing instrument downtime. Before analyzing client samples, the instrument must undergo a tuning process to verify sensitivity, mass resolution, and oxide formation rates. Analysts typically aspirate a dedicated tuning solution containing elements spread across the mass range, such as beryllium, magnesium, cobalt, indium, and lead.
During tuning, technicians must monitor the oxide ratio (commonly measured as the ratio of cerium oxide to cerium, CeO/Ce). If the oxide ratio exceeds the manufacturer's specification—usually 1 to 2 percent—the analyst must adjust the nebulizer gas flow or sampling depth to achieve a hotter, more robust plasma capable of breaking down the sample matrix. Additionally, routine maintenance of the sample introduction system, including regular cleaning of the sampler and skimmer cones, is vital to prevent signal drift caused by salt buildup.
During the analytical run, internal standards are introduced continuously to correct for instrument drift and physical matrix suppression. Internal standards must be elements that are not naturally present in the drinking water samples. Table 2 outlines standard elements used for internal calibration across different mass ranges.
Table 2: Typical Internal Standards for Drinking Water Analysis
Internal Standard | Approximate Mass | Corresponding Analytes Covered |
|---|---|---|
Scandium (Sc) | 45 | Low mass elements (Li, Be, B, Na, Mg) |
Yttrium (Y) | 89 | Mid-low mass elements (Cr, Mn, Fe, Ni, Cu) |
Indium (In) | 115 | Mid mass elements (As, Se, Mo, Ag, Cd) |
Terbium (Tb) | 159 | Mid-high mass elements (Sb, Ba) |
Bismuth (Bi) | 209 | High mass elements (Hg, Tl, Pb, U) |
To maximize throughput, many environmental laboratories implement automated discrete sampling systems. These specialized injection valves push the sample directly into the nebulizer line, bypassing the time-consuming process of pumping the sample through the entire length of the autosampler tubing. This automation can reduce the sample-to-sample analysis time from several minutes to under 60 seconds, drastically increasing lab capacity.
Conclusion: Optimizing ICP-MS operations
Implementing ICP-MS for drinking water analysis provides laboratories with the sensitivity, dynamic range, and efficiency needed to meet increasingly rigorous environmental regulations. By strictly adhering to established sample preparation guidelines, optimizing collision cell technology to remove polyatomic interferences, and automating daily tuning routines, lab managers can maximize both instrument uptime and sample throughput. Ultimately, maintaining highly controlled elemental analysis workflows ensures the delivery of accurate, defensible data that safeguards municipal water supplies and protects public health.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.














