Inductively coupled plasma optical emission spectroscopy (ICP-OES) is a foundational analytical technique for laboratories tasked with material characterization. For laboratory managers overseeing quality control (QC) in advanced manufacturing, the ability to rapidly quantify trace and major elements in complex matrices is critical. Whether verifying the precise composition of a high-performance alloy or detecting trace impurities in semiconductor-grade materials, ICP-OES delivers the dynamic range and high-throughput capabilities required for modern applied science workflows.
Achieving reliable results in these demanding applications requires optimized sample preparation, rigorous spectral interference management, and strict calibration protocols. As industrial material specifications, such as those governed by ASTM International standards, become increasingly stringent, lab leaders must ensure their ICP-OES instruments and analytical methods are finely tuned to handle challenging samples without compromising operational efficiency or data integrity.
Why is ICP-OES essential for alloy and semiconductor characterization?
ICP-OES directly answers the need for simultaneous multi-element analysis across a wide dynamic range. In alloy and semiconductor characterization, laboratories must often measure major matrix components at high percentage levels alongside trace contaminants at parts-per-million (ppm) or parts-per-billion (ppb) levels.
Unlike atomic absorption spectroscopy, which typically analyzes one element at a time, ICP-OES utilizes a high-temperature argon plasma to excite atoms and ions, measuring their specific emission wavelengths simultaneously. This makes it an ideal workhorse for high-throughput QA/QC environments where turnaround time is tightly monitored. Furthermore, the robust nature of the plasma allows the instrument to tolerate high levels of total dissolved solids (TDS), a frequent characteristic of digested metals and highly concentrated semiconductor process chemicals.
How do you overcome sample preparation challenges in complex matrices?
Effective characterization begins well before the sample reaches the plasma; proper matrix digestion is the most critical variable in alloy and semiconductor workflows. These materials are notoriously resistant to standard chemical dissolution. Lab professionals must utilize highly aggressive acid mixtures, often combined with microwave-assisted digestion, to completely break down the solid matrix into a stable, particle-free liquid suitable for nebulization.
For semiconductor applications, trace-level contamination from the digestion process itself must be strictly avoided. Laboratories frequently rely on ultra-high purity (optima-grade) reagents and specialized cleanroom environments to prevent background contamination. For alloys, the primary challenge is keeping high concentrations of elements like tungsten, molybdenum, or titanium in solution without them precipitating prior to analysis. Common digestion approaches are summarized in Table 1.
Table 1: Common Digestion Methods for Alloy and Semiconductor Matrices.
Material Type | Typical Acid Matrix | Primary Operational Challenge |
|---|---|---|
Stainless Steels & Ferrous Alloys | HCl / HNO3 (Aqua Regia) | High iron content creates severe spectral crowding. |
Titanium Alloys | HF / HNO3 | Requires highly specialized HF-resistant sample introduction components. |
Semiconductor Silicon | HF / HNO3 | Highly volatile matrix; risk of analyte loss and background contamination. |
Precious Metals | HCl / HNO3 | Requires precise stoichiometric ratios to prevent analyte precipitation. |
How does ICP-OES mitigate spectral interferences in line-rich samples?
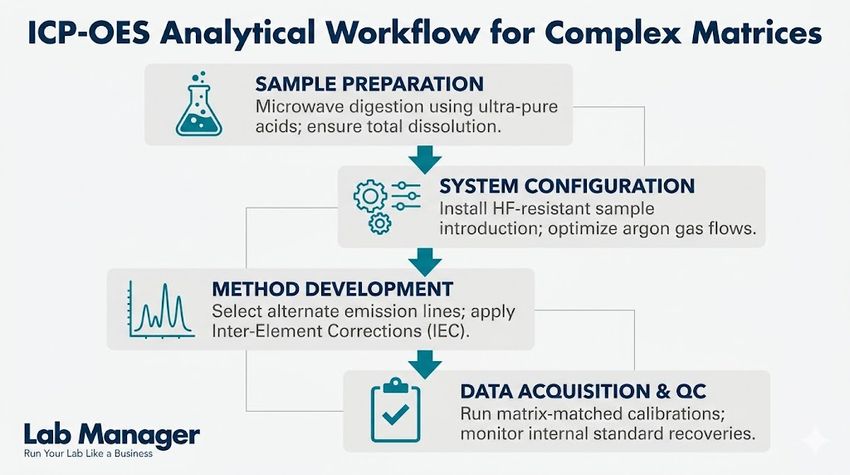
This systematic four-step workflow outlines the essential stages for successful ICP-OES analysis of complex matrices, from rigorous microwave digestion to final quality control validation.
GEMINI (2026)
Spectral interference is the most significant analytical hurdle when running ICP-OES for highly complex matrices. Alloys and semiconductors naturally produce line-rich emission spectra. When a trace element's emission wavelength overlaps with a major matrix element's emission line, it results in false positives or artificially inflated concentration readings.
To combat this, lab managers must ensure standard operating procedures (SOPs) include robust interference mitigation strategies. This often involves selecting alternative, less-interfered analytical wavelengths for trace elements. When alternative lines lack the required sensitivity, analysts rely on inter-element correction (IEC) models or advanced background correction software to mathematically remove the interfering signal. Additionally, matching the acid matrix of NIST-traceable Standard Reference Materials (SRMs) precisely to the sample matrix is an essential step to overcome physical interferences related to sample viscosity and aerosol transport efficiency.
How can labs optimize ICP-OES workflows for high-throughput QA/QC?
In a production-support environment, laboratory efficiency directly impacts the manufacturing supply chain. Lab managers can optimize their ICP-OES workflows by automating routine tasks and minimizing instrument downtime.
Implementing intelligent autosamplers equipped with fast-valve technology can significantly reduce sample uptake and wash-out times, nearly doubling daily sample throughput. Additionally, establishing automated out-of-specification (OOS) flags within the laboratory information management system (LIMS) allows technicians to immediately identify and re-run failing samples without waiting for final batch review. Standardizing reporting formats across both alloy and semiconductor testing lines also ensures that plant managers receive uniform, actionable data regardless of the material being characterized.
What routine maintenance ensures ICP-OES instrument sensitivity?
Because alloy and semiconductor matrices often contain high total dissolved solids and aggressive acids (like hydrofluoric acid), ICP-OES instruments experience accelerated wear on consumable parts. Proactive, preventative maintenance is non-negotiable for reliable characterization.
Lab operations leaders must enforce daily inspections of the sample introduction system. Nebulizers must be checked for partial clogs, which degrade precision, and spray chambers must be monitored for proper droplet formation and drainage. The plasma torch, particularly the injector tube, requires regular cleaning to remove baked-on salt deposits that can cause plasma instability or catastrophic torch melting. By maintaining a strict log of consumable lifespans and replacing parts before analytical performance degrades, labs can maintain the high sensitivity required for trace semiconductor analysis while avoiding unplanned downtime.
Conclusion: Driving quality through rigorous analytical control
For laboratory leaders in advanced manufacturing sectors, mastering ICP-OES for alloy and semiconductor characterization is vital for ensuring product integrity. By addressing the specific challenges of rigorous sample digestion, active spectral interference management, and disciplined instrument maintenance, labs can deliver the accurate, high-throughput elemental data that modern production environments demand. Continuous investment in analyst training and optimized sample handling workflows will ensure that the ICP-OES remains a reliable cornerstone of the QA/QC laboratory.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.














