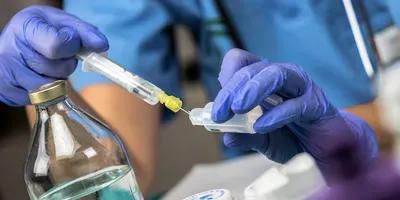
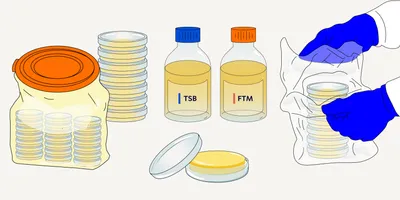
BASEL, SWITZERLAND — May 28, 2020 — Lonza Sales AG, through its Bioscience Division, and Sanquin Reagents B.V. have entered into a strategic partnership for the commercialization of a range of specialized reagents for pyrogen testing of parenteral pharmaceuticals and medical devices using the monocyte activation test (MAT). This partnership expands Lonza’s extensive portfolio of endotoxin testing products, and enables drug developers and quality control (QC) laboratories to explore the full potential of the high-performing, sustainable MAT method to bring safe pharmaceuticals to the market.
“Drug developers and manufacturers are increasingly turning to innovative new technologies that can facilitate accurate, reliable detection of pyrogenic contamination in their products to safeguard patient health," said Archie Cullen vice president, general manager testing & informatics solutions, Lonza. "At Lonza, we are committed to delivering novel solutions that can effectively address this need. By expanding our QC testing portfolio with Sanquin’s state-of-the-art, sustainable MAT kits, we can now provide our customers with a complete toolkit they can rely on to ensure product safety.”
Contrary to the conventional rabbit pyrogen test (RPT), which works by measuring temperature changes in rabbits following administration of a test sample, the MAT does not involve the use of experimental animals. As such, it enables QC laboratories to comply with stringent animal welfare regulations and meet their sustainability objectives. Being a highly sensitive in vitro human cell-based assay, the MAT is capable of consistently detecting pyrogenic activity in even the most complex pharmaceuticals, such as vaccines and cell- and antibody-based biologics, where use of the RPT is limited.
Under the new agreement between Lonza and Sanquin, cryopreserved pooled human peripheral blood mononuclear cells (PBMCs) produced by Sanquin specifically for use with the MAT will bear the Lonza branding. The cells are developed in line with the requirements of the European Pharmacopoeia chapter 2.6.30, following an optimized donor selection, cell isolation and cell quality testing process. Compared to fresh PBMCs, cryopreserved pooled PBMCs eliminate the need to undertake cell isolation for each single test run. As a result, the cells are available on demand whenever a need arises and large number of vials from one batch guarantee a long term test consistency. Lonza will further distribute the Sanquin-branded PeliKine compact ELISA Kits that have been validated for use with the MAT assay. Together with Lonza’s testing hardware, media and accessory offering, drug developers and QC laboratories will now profit from a comprehensive and optimized MAT solution that is in line with regulatory requirements.
“By combining our scientific knowledge and technical expertise on MAT kit production and testing with Lonza's extensive reach and experience with endotoxin testing and data analysis, we will bring the benefits of this powerful technology to a wider audience across the pharmaceutical and biotechnology industries, and further improve the safety of parenteral therapeutics," said Dr. Harry Bos, managing director / CEO, Sanquin Reagents B.V.
For more information about Lonza’s complete testing product offering, please visit https://bioscience.lonza.com/lonza_bs/CH/en/endotoxin-testing For information of Sanquin’s products & services offering, please visit https://www.sanquin.org/mat.