Researchers at the Johns Hopkins Kimmel Cancer Center say they have developed a system that uses transformed human stem cells to speed up screening of existing drugs that might work against rare brain and other cancers.
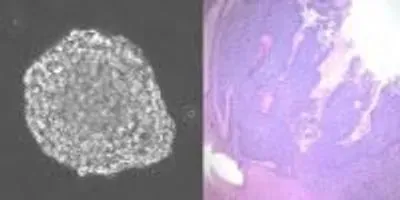
 Left: Neural stem cells transformed with cancer-causing genes found in high-risk medulloblastoma grow as translucent spheres in cell culture (magnification = 200X). Right: Low magnification view of transformed neural stem cells (blue) invading and destroying normal brain tissue (pink) in mice.Image Credit: Allison Hanaford and Eric RaabeA report on their proof-of-concept work, published in the Aug. 1 issue of Clinical Cancer Research, describes experiments that transform human stem cells into an aggressive and rare form of pediatric brain cancer called medulloblastoma. Those cancer cells’ genetic profiles can then be rapidly compared with hundreds of common, lab-grown human cancer cells already tested against existing drugs.
Left: Neural stem cells transformed with cancer-causing genes found in high-risk medulloblastoma grow as translucent spheres in cell culture (magnification = 200X). Right: Low magnification view of transformed neural stem cells (blue) invading and destroying normal brain tissue (pink) in mice.Image Credit: Allison Hanaford and Eric RaabeA report on their proof-of-concept work, published in the Aug. 1 issue of Clinical Cancer Research, describes experiments that transform human stem cells into an aggressive and rare form of pediatric brain cancer called medulloblastoma. Those cancer cells’ genetic profiles can then be rapidly compared with hundreds of common, lab-grown human cancer cells already tested against existing drugs.
By creating a cell model of medulloblastoma from human cells rather than working with mouse cells, the researchers say they can be more confident that patients’ response to the drugs identified in the screenings may be more comparable to humans, according to Eric Raabe, MD, PhD, an assistant professor of oncology at the Johns Hopkins University School of Medicine and a member of the Johns Hopkins Kimmel Cancer Center.
Standard treatment for pediatric medulloblastoma, which strikes 500 children per year in the United States, combines radiation and chemotherapy, but a subtype known as Group 3—which makes up 28 percent of the cases—tends to cause the most relapses in patients, and the survival rate is only 30 to 40 percent, says Raabe. “Overall, 70 to 80 percent of medulloblastoma patients treated with conventional radiation and drug therapy survive and are considered cured,” he adds, “but many patients with the Group 3 subtype don’t fare as well.”
In recent years, one strategy scientists use to find new or better treatments is to look to databases of existing drugs that have been matched to the genetic profiles of lab-grown cells for common cancers. However, Raabe says, rare cancers and their subtypes are not well-represented in such databases, and creating such lab-grown cell lines directly from patients’ tumors is difficult. Moreover, he adds, such tumor-derived cells sometimes acquire genetic changes that can vary from the original tumor.
For the study, the scientists used lentiviruses as a transport system to insert cancer-related genes common to Group 3 medulloblastomas in human neural stem cells. As the stem cells replicate, the cancer-linked genes transform the stem cells into cancer cells. The tumors then grown from these transformed cell lines “compare very well biologically to actual human medulloblastoma, and their gene expression profile fits with that of human tumor cells,” Raabe notes.
Raabe and his colleagues then used RNA sampled from the new tumors to create a “signature” of gene expression that they could compare to similar signatures in three large databases of cell lines that have been screened and matched with existing drugs. “We wanted to find whether the cells we created matched any of these existing signatures,” Raabe explains, “because if they did, then we would have some idea of what kinds of drugs are more or most likely to kill these cells. We didn’t have to do the laborious screening to test 100,000 compounds against our own cells.”
Using this method, the scientists zeroed in on a group of compounds called CDK inhibitors that may be promising treatments for Group3 medulloblastoma, Raabe says.
One of those drugs, palbociclib, is already approved by the U.S. Food and Drug Administration for treating a type of advanced breast cancer, Raabe says. When he and his colleagues added palbociclib to their transformed cell lines, they found that the drug decreased cell line growth by more than 50 percent and more than tripled cell death compared to untreated cells. The drug also extended the survival of mice implanted with tumors grown from established Group 3 human medulloblastoma cell lines by nearly 50 percent, from 25 to 37 days.
Related Article: Turning Biological Cells to Stone Improves Cancer and Stem Cell Research
Raabe says that palbociclib and other such drugs, called cyclin-dependent kinase inhibitors, are being tested in phase I clinical trials in children with various brain tumors. “There is interest in testing these agents in more advanced studies specifically for recurrent medulloblastoma, potentially in combination with other new agents,” he says.
Although it took several years to develop this model of Group 3 medulloblastoma and show its similarity to primary tumors, Raabe says, “This system may be one way to find drugs for rare cancers for which there are only a few human cell lines or to model very rare subtypes of adult and pediatric cancers.”
Other scientists who contributed to the research include Allison Hanaford, Antoinette Price and Charles Eberhart at Johns Hopkins; Tenley C Archer, Jong Wook Kim, Tobias Ehrenberger, Paul A. Clemons, Vlado Daník, Brinton Seashore-Ludlow, Vasanthi Viswanathan, Michelle L Stewart, Matthew G. Rees, Alykhan Shamji, Stuart Schreiber, Ernest Fraenkel, Scott L. Pomeroy, Jill P. Mesirov, and Pablo Tamayo of the Broad Institute at MIT and Harvard; Ulf D. Kahlert and Jarek Maciaczyk of Heinrich-Heine University Hospital Duesseldorf, Germany; and Guido Nikkhah of University Hospital Stuttgart, Germany.
Funding for the study was provided by the St. Baldrick’s Foundation, Hyundai Hope on Wheels, Giant Food’s Pediatric Cancer Research Fund, the Spencer Grace Foundation, the Deming Family, the Children’s Brain Tumor Foundation, the National Cancer Institute (P30 CA006973, R01NS055089, U01CA176152), the National Institutes of Health (R01 CA154480, R01 109467, R01GM074024), and the Comprehensive Cancer Center Freiburg.