For pharmaceutical quality control (QC) managers, ensuring compliance with ICH Q3D standards is a critical operational requirement. These guidelines mandate the rigorous assessment and control of elemental impurities in drug products to guarantee patient safety and product efficacy.
Inductively coupled plasma optical emission spectroscopy (ICP-OES) has emerged as a workhorse technique for this application, offering a balance of sensitivity, high throughput, and cost-effectiveness. Establishing an efficient ICP-OES workflow allows laboratories to confidently meet regulatory thresholds while managing routine testing volumes and operational budgets.
What are the ICH Q3D elemental impurity classes?
The International Council for Harmonisation (ICH) Q3D guideline classifies elemental impurities into three main categories based on their toxicity and likelihood of occurrence in the pharmaceutical manufacturing process. Understanding these classes is the first step in developing a targeted, risk-based testing strategy for your lab.
- Class 1: Elements like arsenic (As), cadmium (Cd), mercury (Hg), and lead (Pb) are highly toxic to humans and have limited or no use in pharmaceutical manufacturing. They require evaluation during the risk assessment across all potential sources.
- Class 2: These elements are considered route-dependent human toxicants, meaning their permitted daily exposure (PDE) limits differ significantly depending on whether the drug is administered orally, parenterally, or via inhalation. They are further divided into Class 2A (relatively high probability of occurrence, requiring assessment) and Class 2B (reduced probability of occurrence, requiring assessment only if intentionally added during manufacturing).
- Class 3: Elements with relatively low toxicities by the oral route of administration. Class 3 elements have higher PDEs for oral routes but may require tighter control for parenteral or inhalation products. They typically only require evaluation if intentionally added.
Table 1: ICH Q3D Elemental Impurity Classifications
Class | Elements | Risk Assessment Requirement |
|---|---|---|
Class 1 | As, Cd, Hg, Pb | Always required |
Class 2A | Co, Ni, V | Always required |
Class 2B | Ag, Au, Ir, Os, Pd, Pt, Rh, Ru, Tl | Required if intentionally added |
Class 3 | Ba, Cr, Cu, Li, Mo, Sb, Sn | Required if intentionally added |
Why choose ICP-OES for pharmaceutical elemental analysis?
ICP-OES provides a highly efficient analytical platform for trace metal analysis, particularly for laboratories processing high volumes of pharmaceutical samples. While inductively coupled plasma mass spectrometry (ICP-MS) offers superior sensitivity and lower detection limits, ICP-OES remains highly attractive for lab managers due to its lower initial capital investment, reduced operational complexity, and lower consumable costs.
For many pharmaceutical products, the permitted daily exposure (PDE) limits established by ICH Q3D translate into target concentrations that fall comfortably within the detection range of modern ICP-OES instruments. Furthermore, ICP-OES systems generally exhibit higher tolerance for total dissolved solids (TDS), making them highly robust for analyzing complex, heavy-matrix pharmaceutical formulations without the frequent maintenance downtime associated with ICP-MS interface cones.
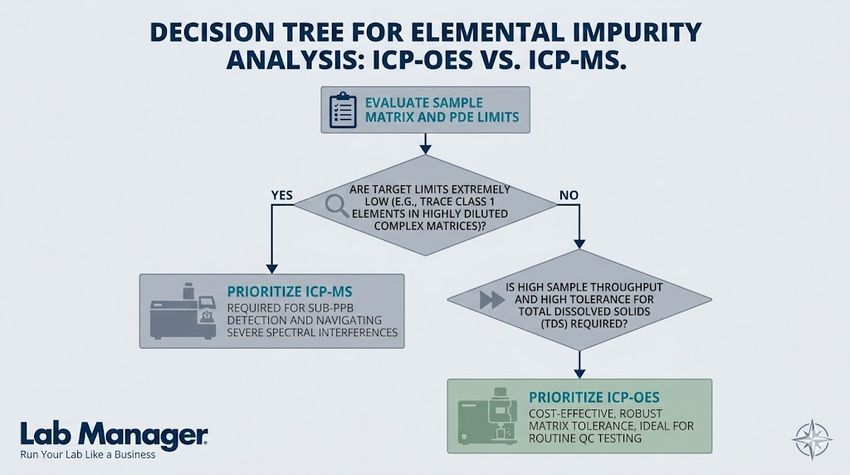
Use this flowchart to determine whether your lab's elemental impurity testing requires the high sensitivity of ICP-MS or if the robust matrix tolerance of ICP-OES is sufficient.
GEMINI (2026)
Sample preparation strategies for ICP-OES analysis
Effective sample preparation is the most critical step in achieving accurate ICP-OES results for ICH Q3D compliance. Because pharmaceutical samples range from simple aqueous solutions to complex lipid-based suspensions and solid oral dosages, the preparation method must ensure complete dissolution of the target analytes without introducing external contamination.
Microwave-assisted acid digestion is the industry standard for solid and semi-solid pharmaceutical samples, aligning with the sample preparation guidelines outlined in USP <233>. Using closed-vessel microwave digestion ensures the complete breakdown of complex organic matrices while preventing the loss of volatile elemental impurities, such as mercury and arsenic. Laboratories must utilize high-purity, trace-metal-grade reagents (such as nitric and hydrochloric acids) to keep method blank concentrations well below the limit of quantitation (LOQ).
ICP-OES instrument calibration and USP <233> validation protocols
Validating an ICP-OES method for ICH Q3D compliance requires demonstrating specificity, accuracy, precision, and an appropriate LOQ, typically aligned with USP <233> validation requirements. To manage matrix effects—which are common when analyzing dissolved pharmaceutical products—laboratories frequently employ internal standardization.
Yttrium or scandium are commonly added as internal standards to correct for physical interferences, such as differences in sample viscosity or aerosol transport efficiency. Furthermore, careful selection of emission wavelengths is necessary to avoid spectral interferences from the sample matrix or from other elements present in the formulation. Routine system suitability checks, including the analysis of continuous calibration verification (CCV) standards, ensure the instrument maintains its performance throughout long analytical runs.
Conclusion: Optimizing ICP-OES workflows for ICH Q3D compliance
Implementing a compliant ICH Q3D workflow using ICP-OES requires laboratory leaders to balance instrument capabilities, rigorous sample preparation, and strict adherence to validation protocols. By understanding the specific PDE limits required for their products and optimizing their microwave digestion and analytical methods, lab managers can leverage ICP-OES as a robust, cost-effective solution for elemental impurity testing. Proper training, preventative maintenance, and method optimization will ensure the laboratory consistently delivers accurate data, safeguarding both product quality and operational efficiency.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.














