For quality assurance (QA) and quality control (QC) managers, raw material identification is a high-volume, mandatory process that often creates critical bottlenecks in the supply chain. Before any manufacturing process can begin—whether in pharmaceuticals, food and beverage, or specialty chemicals—incoming raw materials must be definitively identified to prevent costly production errors and ensure consumer safety. Historically, this required time-consuming wet chemistry techniques, hazardous solvent extraction, and complex chromatographic analysis, all of which consume the sample and drive up the cost per test.
To alleviate these operational burdens, laboratory leaders are widely transitioning to non-destructive identity testing. By implementing advanced spectroscopic technologies, primarily Fourier-transform infrared (FTIR) spectroscopy and near-infrared (NIR) spectroscopy, QA facilities can dramatically reduce turnaround times. These optical techniques allow technicians to verify chemical identities in seconds rather than hours, without destroying the raw material or generating hazardous chemical waste. Understanding the distinct operational advantages and limitations of FTIR and NIR is essential for lab managers looking to optimize their high-throughput QA workflows.
What is non-destructive identity testing?
Non-destructive identity testing refers to analytical methodologies that verify the chemical composition of a substance without permanently altering or consuming the sample. In a traditional QA workflow, a sample is pulled from the warehouse, transported to the lab, weighed, dissolved in chemical solvents, and analyzed using techniques like high-performance liquid chromatography (HPLC) or titration. This destructive process destroys the tested aliquot, requires extensive consumable usage, and creates hazardous waste that the facility must pay to dispose of safely.
Conversely, non-destructive techniques rely on the interaction between light and matter. When a material is exposed to specific wavelengths of infrared light, its molecular bonds absorb the energy, creating a unique spectral "fingerprint." Because this process only involves light exposure, the sample remains physically and chemically intact. For lab managers, the operational benefits are immediate: reduced consumable spending, elimination of solvent disposal costs, and a drastic reduction in the labor hours required for sample preparation. Furthermore, many non-destructive instruments can be operated by technicians with basic training, freeing up highly skilled analytical chemists to focus on complex troubleshooting and method development.
How does FTIR streamline raw material identification?
Fourier-transform infrared (FTIR) spectroscopy operates in the mid-infrared region of the electromagnetic spectrum. It is highly sensitive to the fundamental vibrations of molecular bonds, making it an exceptionally powerful tool for identifying pure organic compounds, polymers, and certain inorganic materials. For modern QA laboratories, the introduction of Attenuated Total Reflectance (ATR) accessories has revolutionized FTIR workflows.
ATR-FTIR allows for the direct analysis of solid powders, pastes, and liquids with zero sample preparation. A technician simply places a small amount of the sample directly onto a durable crystal (typically diamond or zinc selenide) and applies pressure. The instrument measures the infrared beam as it internally reflects through the crystal and interacts with the sample surface.
In a high-throughput QA environment, FTIR offers several distinct operational advantages:
- Speed: A complete identity scan and library match can be executed in less than a minute.
- Specificity: Mid-infrared spectra provide highly detailed structural information, easily distinguishing between closely related chemical analogs or structural isomers.
- Ease of cleaning: The ATR crystal is easily wiped clean with a solvent-dampened cloth between analyses, preventing cross-contamination and maximizing instrument uptime.
However, lab managers must note that ATR-FTIR is fundamentally a surface analysis technique; the light only penetrates a few micrometers into the sample. Therefore, the sample must be highly homogeneous. Additionally, because mid-infrared light is strongly absorbed by glass and plastics, FTIR typically requires the sample to be physically removed from its primary packaging prior to analysis.
When should your lab choose NIR for QA workflows?
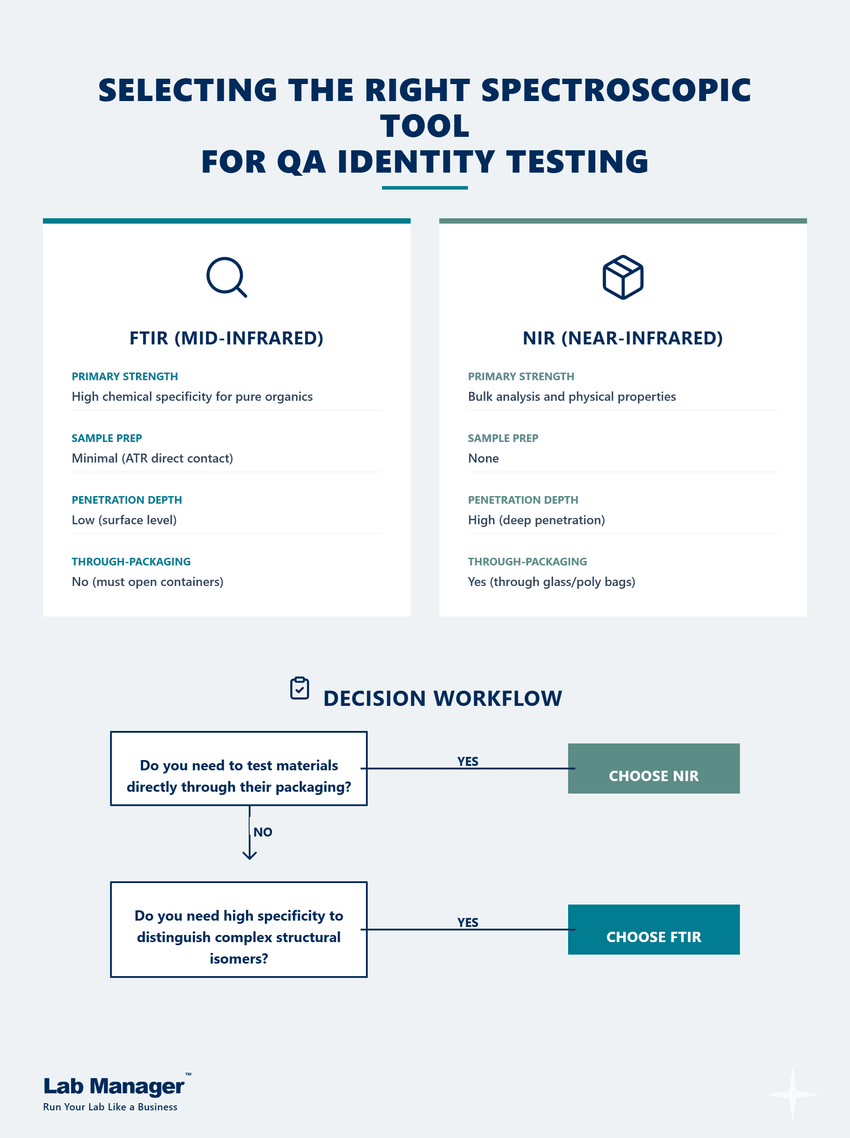
Use this quick reference guide to compare their primary strengths and choose the right spectroscopic tool based on your packaging and chemical specificity requirements.
GEMINI (2026)
Near-infrared (NIR) spectroscopy operates at higher frequencies than FTIR, analyzing the overtones and harmonic vibrations of molecular bonds (primarily C-H, N-H, and O-H bonds). While NIR spectra are broader and visually less distinct than the sharp peaks seen in FTIR, NIR light penetrates much deeper into samples. This fundamental difference makes NIR a powerhouse for bulk material analysis and continuous process monitoring, often forming the foundation of Process Analytical Technology (PAT) frameworks advocated by regulatory agencies.
For QA operations, the greatest advantage of NIR is its ability to perform non-destructive identity testing directly through transparent or translucent packaging, such as glass vials or polyethylene bags. By utilizing fiber-optic probes or handheld NIR analyzers, QA personnel can verify raw materials directly on the receiving dock or in the warehouse. This eliminates the need to transport materials to a central laboratory, open sealed containers, or expose operators to potentially hazardous active pharmaceutical ingredients (APIs).
NIR is highly recommended for lab workflows involving:
- High-volume warehouse receiving: Rapid verification of hundreds of incoming drums without opening sterile seals.
- Moisture analysis: NIR is exceptionally sensitive to water, allowing simultaneous identity verification and moisture content quantification.
- Physical property assessment: Unlike FTIR, NIR is sensitive to physical characteristics such as particle size and crystalline form, providing broader quality metrics beyond simple chemical identity.
Overcoming implementation and compliance challenges in QA
While the operational benefits of non-destructive identity testing are substantial, laboratory managers must carefully navigate the implementation phase to ensure regulatory compliance and data integrity. The effectiveness of both FTIR and NIR is entirely dependent on the quality of the spectral libraries used for comparison.
Unlike some analytical techniques that provide absolute quantitative values, spectroscopic identity testing relies on chemometrics—the application of mathematical and statistical methods to chemical data. The instrument compares the spectrum of the incoming raw material against a validated reference library of known standards. Developing these robust libraries requires significant upfront labor. Lab managers must ensure their teams scan multiple batches of raw materials from approved suppliers, accounting for acceptable natural variations in moisture, particle size, and trace impurities. If the library is too narrow, the system will generate false failures, rejecting perfectly good materials and halting production.
Furthermore, heavily regulated industries, such as pharmaceuticals, mandate strict adherence to data integrity frameworks, notably 21 CFR Part 11 in the United States. When selecting FTIR or NIR instrumentation, lab managers must verify that the accompanying software includes unalterable audit trails, secure user access controls, and electronic signature capabilities. The methodology itself must also be validated according to pharmacopeial standards, such as USP <856> for near-infrared, USP <854> for mid-infrared, and USP <858> for Raman spectroscopy. Investing in comprehensive vendor training and user-friendly software is critical to minimizing implementation downtime and ensuring defensible QA data during regulatory audits.
Conclusion: Elevating QA efficiency through spectroscopic technologies
Transitioning to non-destructive identity testing represents a strategic upgrade for any quality assurance laboratory facing high sample volumes and strict turnaround requirements. By integrating FTIR and NIR into routine testing workflows, lab managers can drastically reduce the cost per sample, eliminate hazardous solvent consumption, and reallocate highly trained personnel to more complex analytical tasks. While the initial investment in instrumentation and chemometric library building requires careful planning, the long-term operational efficiency and improved supply chain velocity make spectroscopic identity testing an indispensable tool for modern, compliant laboratory operations.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.














