Analytical laboratories require rigorous adherence to proper storage, maintenance, and pH meter electrodes protocols to guarantee the accuracy, repeatability, and reliability of potentiometric measurements. Neglecting these established procedures rapidly degrades the sensitive glass membrane and the reference junction, instantly compromising data integrity across critical laboratory assays. Implementing standardized care routines minimizes baseline drift, accelerates response times, and significantly extends the operational lifespan of the analytical sensor.
What are the proper storage conditions for pH meter electrodes?
The structural integrity of a pH sensor relies on continuous hydration of the silicate glass membrane to maintain an active, conductive ion-exchange layer. When left exposed to ambient air, this delicate gel layer dehydrates, causing erratic voltage readings, sluggish response times, and total sensor failure. Proper storage, maintenance, and pH meter electrodes practices require immersing the glass bulb in a designated electrolytic storage solution to preserve this chemically active boundary.
Analytical pH glass is intentionally manufactured with a porous microscopic structure that interacts seamlessly with free hydrogen ions in the target sample. This chemical interaction creates the millivolt potential read by the analytical meter, a process that completely halts if the delicate glass dries out. Rehydrating a severely desiccated membrane requires extensive soaking in acidic solutions, though prolonged dehydration often causes irreversible structural micro-fissures in the glass.
Storage solutions also maintain the vital electrochemical balance of the internal reference system, which is typically composed of a silver/silver chloride (Ag/AgCl) element. Submerging the sensor in an appropriate molarity of potassium chloride (KCl) prevents the internal electrolyte from leaching out through the porous ceramic junction, a critical standard upheld by the United States Pharmacopeia (USP). If the internal reference electrolyte becomes depleted due to improper storage conditions, the electrical circuit breaks, rendering the electrode permanently inoperable.
Why storing pH meter electrodes in deionized water causes sensor failure
Storing pH electrodes in deionized (DI) or reverse osmosis (RO) water actively extracts internal electrolyte ions through the reference junction via osmotic pressure differentials. Because DI water contains virtually zero dissolved ions, it creates a severe concentration gradient that forces potassium and chloride ions out of the sensor body. This rapid depletion of the internal reference solution drastically shortens the lifespan of the equipment and necessitates frequent, costly sensor replacements.
In addition to rapid electrolyte depletion, DI water storage causes the silver chloride complex within the reference element to precipitate and crystallize. These solid silver chloride crystals physically lodge within the microscopic pores of the ceramic or Teflon reference junction, creating an insulating physical barrier. Once the junction becomes blocked by these precipitates, the electrode exhibits severe baseline drift and a complete inability to stabilize during routine measurements.
To avoid these catastrophic equipment failures, standard analytical methods from ASTM International strictly recommend using a 3M or 4M KCl solution for routine storage. Commercially prepared electrode storage solutions are specifically formulated to match the internal electrolyte concentration exactly, thereby neutralizing any destructive osmotic pressure. Adhering to these established storage, maintenance, and pH meter electrodes guidelines ensures the reference junction remains clear, fully conductive, and free of physical blockages.
Operators must carefully train all laboratory personnel to recognize the visual differences between standard cleaning wash bottles and long-term chemical storage vessels. A common operational error involves technicians inadvertently topping off sensor storage caps with pure laboratory water rather than the required ionic KCl solution. Instituting color-coded reagent protocols dramatically reduces these human errors and protects the sensitive internal chemistry of the high-precision analytical instruments.
Standardized chemical maintenance protocols for pH meter electrodes
Standardized maintenance protocols dictate that pH electrodes must be cleaned with specific chemical agents based directly on the precise nature of the sample contaminant. Routine rinsing with distilled water effectively removes simple water-soluble salts, but it is entirely ineffective against complex biological molecules, oils, or inorganic precipitates. Implementing targeted chemical cleaning restores the sensor's baseline performance and prevents the accumulation of insulating boundary layers on the delicate glass bulb.
Proteinaceous buildup from biological samples requires targeted enzymatic cleaning to break down complex peptide bonds that adhere strongly to the glass membrane. Laboratories typically utilize a 0.1 M hydrochloric acid (HCl) solution containing 1% pepsin, soaking the electrode for one to two hours to digest the proteins. Failing to remove protein coatings leads to sluggish response times, as the hydrogen ions in the sample cannot physically reach the active silicate gel layer.
Inorganic foulants, particularly silver sulfide precipitates, demand a completely different chemical approach to effectively restore reference junction permeability and baseline stability. A solution of 0.1 M HCl heavily saturated with thiourea effectively dissolves these dark precipitates, clearing the reference junction and restoring electrical continuity. For heavy oil and lipid contamination, gentle washing with a mild, non-ionic surfactant safely removes the hydrophobic film without etching the sensitive glass.
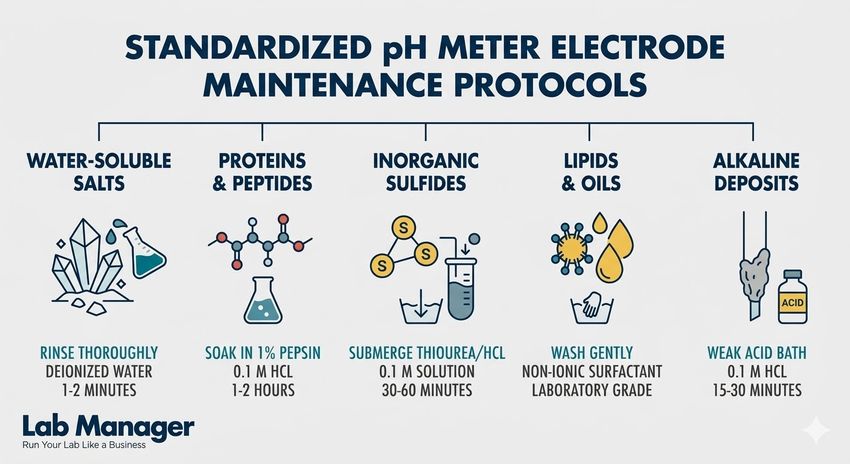
A quick reference guide to standardized maintenance protocols for cleaning pH meter electrodes based on specific laboratory contaminants.
GEMINI (2026)
- Water-soluble salts: Rinse thoroughly with deionized water for 1-2 minutes.
- Proteins and peptides: Soak in 1% pepsin within 0.1 M HCl for 1-2 hours.
- Inorganic sulfides: Submerge in 0.1 M thiourea/HCl solution for 30-60 minutes.
- Lipids and oils: Wash gently with a laboratory-grade non-ionic surfactant.
- Alkaline deposits: Soak in a weak 0.1 M hydrochloric acid bath for 15-30 minutes.
Following any aggressive chemical cleaning procedure, the electrode must be thoroughly rinsed and soaked in standard storage solution to reestablish the hydration layer. Harsh chemical agents physically strip the delicate gel boundary on the glass bulb, rendering the sensor temporarily unresponsive until the layer successfully rebuilds. This mandatory recovery period ensures that the subsequent calibration cycle accurately reflects the sensor's true potentiometric capabilities and optimal slope.
How internal reference systems dictate pH meter electrode maintenance
The physical architecture of the internal reference system directly determines the specific storage and maintenance procedures required for a given analytical pH electrode. Liquid-filled electrodes feature a refillable outer chamber that allows operators to physically replenish the internal electrolyte solution as it naturally drains through the junction. This design necessitates routine visual inspections of the fill hole and regular top-offs with appropriate KCl solutions to maintain adequate internal hydrostatic pressure.
If the internal liquid level falls below the exterior sample level, reverse flow occurs, drawing potentially hazardous or contaminating sample fluid into the electrode body. Maintaining proper hydrostatic pressure by keeping the fill hole open during use and closed during storage prevents this catastrophic internal fluid contamination. Operators must strictly follow these mechanical storage, maintenance, and pH meter electrodes procedures to protect the delicate internal silver wire from irreversible chemical poisoning.
Conversely, gel-filled electrodes utilize a highly viscous polymer matrix to suspend the reference electrolyte, completely eliminating the need for manual liquid refilling. While these low-maintenance sensors offer substantial operational convenience, they possess a finite lifespan because the internal gel cannot be replenished once depleted. When a gel-filled electrode exhibits sluggish responses due to natural electrolyte exhaustion, no maintenance procedure can reverse the degradation, and the unit requires immediate replacement.
How proper physical handling extends pH meter electrode lifespan
Proper physical handling requires laboratory operators to blot the glass membrane gently with a lint-free tissue rather than wiping it aggressively. Wiping the sensitive glass bulb generates a localized static electrical charge that violently disrupts the sensor's voltage reading and causes prolonged signal instability. This static interference can take several minutes to dissipate entirely, significantly slowing down high-throughput laboratory workflows and introducing temporary measurement errors.
Furthermore, aggressive mechanical wiping physically abrades the microscopic hydrated gel layer on the exterior of the sensitive silicate glass over time. Repeated mechanical degradation of this vital boundary layer permanently alters the sensor's slope and asymmetry potential, leading directly to inevitable calibration failures. Gentle blotting effectively removes excess moisture between individual sample readings without inducing static polarization or damaging the essential ion-exchange surface.
Following guidelines published by the World Health Organization (WHO) regarding good laboratory practices ensures that these high-precision instruments survive daily use. Operators must always secure electrodes in dedicated mechanical articulating arms when taking measurements to prevent accidental, damaging impacts with the beaker. The thin glass bulb is highly susceptible to mechanical shock, and even microscopic fractures completely destroy the unit's ability to hold a millivolt potential.
Proper calibration frequency and buffer selection: Proper calibration frequency and buffer selection are fundamental prerequisites for translating raw millivolt potentials into accurate, actionable laboratory pH values. Standard operating procedures dictate that analytical meters must be calibrated daily using fresh, NIST-traceable buffer solutions that carefully bracket the expected pH range of the target samples. By ensuring buffers are stored at ambient room temperature and never reused across different calibration cycles, laboratories maintain the rigorous metrological traceability required by global regulatory bodies.
Conclusion: Maximizing lifespan through proper storage, maintenance, and pH meter electrodes protocols
Implementing rigorous and standardized care protocols is non-negotiable for maintaining the precise analytical capabilities of modern laboratory instrumentation. Adhering directly to these optimized storage, maintenance, and pH meter electrodes procedures guarantees accurate data generation, prevents reference junction clogging, and significantly extends the operational lifespan of the sensor. By strictly eliminating physical mishandling and deliberately avoiding improper reagents like deionized water, analytical laboratories can confidently ensure sustained metrological reliability across all workflows.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.