Accurate determination of acidity in processed foods with titrators is a fundamental requirement for verifying product safety, extending shelf life, and meeting strict regulatory compliance. Laboratory professionals rely on these automated analytical instruments to quantify titratable acidity, a critical metric that dictates microbial stability and flavor consistency in commercial food manufacturing. Utilizing standardized titration protocols ensures that complex food matrices, ranging from emulsified dairy products to canned vegetables, meet the stringent quality control benchmarks established by global food safety organizations.
How do titrators measure titratable acidity in processed foods?
Titrators measure titratable acidity in processed foods by incrementally adding a standard base solution of known concentration to a food sample until a specific endpoint is reached, neutralizing the available organic acids. This process relies on fundamental acid-base chemical reactions where the titrant reacts with the hydrogen ions present in the food matrix. The volume of the titrant required to reach the neutralization point is then used to calculate the total concentration of the predominant acid in the sample.
In modern laboratory environments, potentiometric titrators replace manual visual indicators by utilizing highly sensitive pH electrodes to detect the exact equivalence point of the chemical reaction. This automated approach is essential because many processed foods, such as dark fruit juices or thick tomato pastes, are opaque or heavily colored. The intrinsic color of these food matrices makes it impossible to accurately observe the color changes associated with traditional chemical indicators like phenolphthalein.
To determine the final acidity profile, the instrument's microprocessor calculates the result based on the specific organic acid most prevalent in the food product. Food scientists must configure the instrument to express the results using the appropriate acid coefficient to ensure standardized reporting. Common predominant acids analyzed in processed foods include:
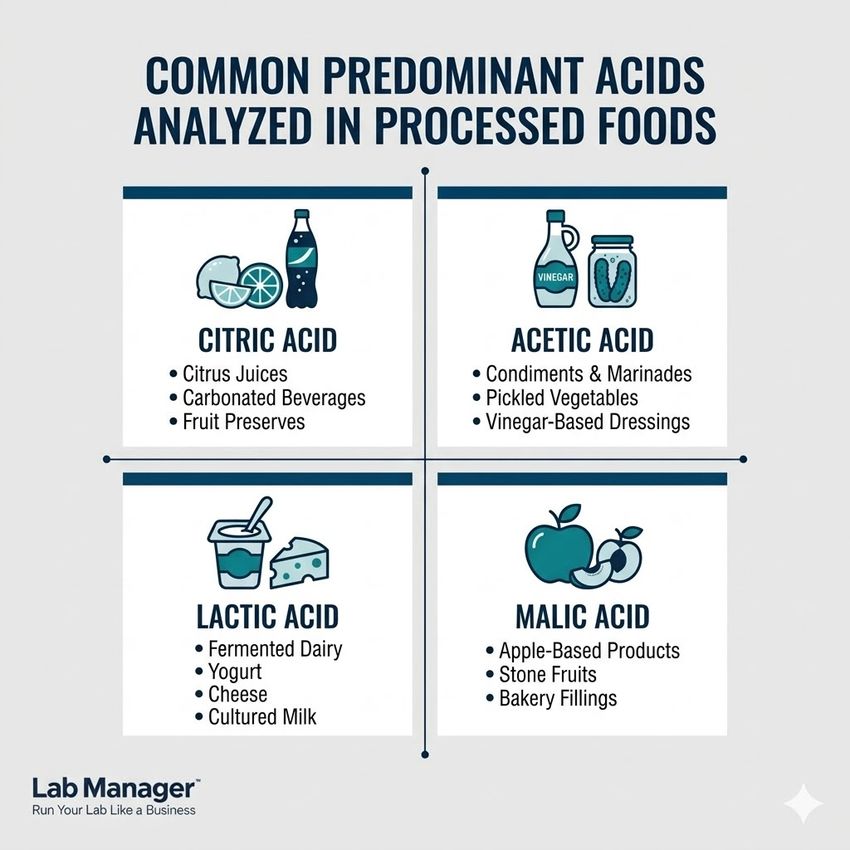
A Quick Guide to Organic Acids in Food Processing: This breakdown highlights the four predominant acids—citric, acetic, lactic, and malic—commonly analyzed in processed foods, alongside their primary product applications.
GEMINI (2026)
- Citric acid: The primary reference acid used when titrating citrus juices, carbonated beverages, and fruit-based preserves.
- Acetic acid: The standard reference used for evaluating condiments, marinades, pickled vegetables, and vinegar-based dressings.
- Lactic acid: The critical measurement standard for fermented dairy products, including yogurt, cheese, and cultured milk.
- Malic acid: The primary organic acid evaluated when profiling apple-based products, stone fruits, and specific bakery fillings.
The precision of modern titrators allows laboratories to measure these acids down to trace levels, ensuring absolute consistency across massive commercial production batches. By automating the dosing of the titrant, these devices eliminate human error related to manual burette reading and inconsistent dispensing speeds. Consequently, analytical chemists can achieve highly reproducible acidity profiles even when processing hundreds of samples per day.
Why is distinguishing between total acidity and pH critical for food safety?
Distinguishing between titratable acidity and pH is critical for food safety because pH measures only the free hydrogen ion concentration, whereas titratable acidity quantifies the total buffer capacity and total organic acid content of the food matrix. Both metrics provide distinct, essential data points regarding the chemical stability and safety profile of the final product. Relying solely on one measurement can lead to catastrophic failures in food preservation and regulatory compliance.
The pH level acts as the primary indicator for inhibiting the growth of dangerous pathogens, most notably Clostridium botulinum, which produces deadly botulinum toxin. According to the United States Food and Drug Administration (FDA) regulations outlined in 21 CFR Part 114, acidified processed foods must maintain an equilibrium pH of 4.6 or lower to prevent the germination of these harmful spores. While pH ensures immediate microbial safety, it does not provide a complete picture of how the food will taste or how it will interact with other ingredients over its shelf life.
Conversely, measuring total acidity in processed foods with titrators provides vital insights into the product's organoleptic properties, specifically its perceived tartness and flavor profile. Two distinct food products can exhibit the exact same pH level but possess vastly different titratable acidity values due to varying buffer capacities within their ingredients. If a food manufacturer only monitors pH, they risk producing a safe product that is unpalatable to the consumer due to overwhelming sourness.
Therefore, comprehensive laboratory analysis requires the simultaneous evaluation of both parameters to ensure a product is both biologically safe and commercially viable. Automated titrators frequently offer combined pH and titratable acidity modes, allowing technicians to capture both data points from a single sample aliquot. This dual-analysis approach is widely recognized as a best practice by organizations such as AOAC International and the World Health Organization (WHO) for establishing robust food safety frameworks.
What are the primary types of titrators used for food laboratory analysis?
The primary types of titrators used for food laboratory analysis include manual volumetric burettes, potentiometric titrators, and fully automated multi-parameter titration systems. The selection of the appropriate instrument depends entirely on the sample throughput requirements, the complexity of the food matrices being analyzed, and the necessary degree of analytical precision. While traditional manual titration remains an acceptable educational tool, commercial food testing laboratories almost exclusively utilize electronic systems.
Potentiometric titrators are the standard instruments used for analyzing acidity in processed foods, utilizing a glass pH electrode to continuously monitor the sample's potential difference as the titrant is added. These devices mathematically determine the equivalence point by analyzing the first and second derivatives of the titration curve, eliminating the subjectivity of visual endpoint detection. This technology is highly adaptable and can be configured with automated autosamplers to process dozens of continuous samples without human intervention.
Thermometric titrators represent another advanced category, operating on the principle that acid-base neutralization reactions are inherently exothermic. Instead of measuring electrical potential, these instruments utilize highly responsive thermistors to detect minute temperature spikes that occur precisely at the chemical endpoint. Thermometric titration is exceptionally advantageous when analyzing high-fat processed foods, such as mayonnaise or creamy salad dressings, which rapidly foul the porous junctions of traditional glass pH electrodes.
To optimize equipment selection, laboratory managers must evaluate the specific capabilities and operational limitations of each technology:
Titrator Classification | Primary Mechanism of Action | Ideal Food Matrix Applications | Operational Limitations |
|---|---|---|---|
Manual Volumetric | Visual observation of color-changing chemical indicators. | Clear liquids, refined waters, simple brines. | Highly subjective; unusable for colored or opaque samples. |
Potentiometric | Electrochemical detection of hydrogen ion concentration. | Juices, sauces, wines, dairy products, canned vegetables. | Electrodes require frequent calibration and careful maintenance. |
Thermometric | Detection of enthalpy changes (temperature) during reaction. | High-fat foods, heavy emulsions, viscous dairy, thick pastes. | Requires specific sensor probes and controlled ambient temperatures. |
Transitioning from manual to automated titrators significantly enhances laboratory throughput while reducing the chemical waste generated by repeated, erroneous manual trials. Furthermore, modern automated systems integrate directly with Laboratory Information Management Systems (LIMS), ensuring that all generated data is instantly archived and traceable.
How do sample preparation techniques affect acidity profiles in complex matrices?
Sample preparation techniques directly affect acidity profiles in complex food matrices by altering the solubility, homogeneity, and accessibility of organic acids prior to titration. If a food sample is improperly prepared, the titrant cannot effectively interact with the internal cellular structures of the food, resulting in artificially low acidity readings. Establishing rigorous, standardized sample preparation protocols is just as critical as the calibration of the titration equipment itself.
For solid and semi-solid processed foods, such as baked goods, processed meats, or fruit preserves, thorough homogenization is an absolute requirement. Laboratory technicians must use high-shear blenders or stomachers to physically break down the food matrix into a uniform paste or slurry. This process usually involves blending the sample with a specific, accurately weighed volume of distilled or deionized water to extract the organic acids into an aqueous phase.
Carbonated processed foods, including sodas, sparkling waters, and fermented kombuchas, require immediate degassing prior to any acidity analysis. Dissolved carbon dioxide reacts with water to form carbonic acid, which acts as an active acid during the titration process and severely inflates the titratable acidity results. Technicians must remove the carbon dioxide through prolonged ultrasonic agitation, vacuum filtration, or gentle heating before introducing the sample to the titrator.
Handling emulsions and high-fat matrices presents a distinct set of challenges that require specialized solvent extraction techniques. Fats can coat the pH electrodes of potentiometric titrators, physically blocking the sensor and causing erratic, drifting readings. In these instances, laboratories often utilize non-aqueous titration methods or introduce specific surfactant solvents to break the emulsion and ensure the acids are fully accessible to the titrant.
What quality control protocols ensure accuracy when using titrators for food analysis?
Implementing rigorous quality control protocols, including daily electrode calibration and standardized titrant verification, ensures accuracy and reproducibility when using titrators for food analysis. Without a strict adherence to routine maintenance and standardization, the inherent drift of electronic sensors will inevitably lead to compromised analytical data. Regulatory bodies, including the FDA and ISO, mandate that laboratories maintain comprehensive audit trails documenting these daily quality control procedures.
The cornerstone of titration accuracy is the multi-point calibration of the pH electrode using certified National Institute of Standards and Technology (NIST) traceable buffer solutions. Laboratory professionals must calibrate the electrode daily, typically utilizing pH 4.01, 7.00, and 10.01 buffers, to establish a precise slope and offset for the sensor. If the electrode's slope falls below 95 percent of the theoretical Nernstian value, the probe must be reconditioned or immediately replaced to prevent inaccurate endpoint detection.
Equally important is the routine standardization of the titrant solution, usually sodium hydroxide (NaOH), which can degrade or absorb atmospheric carbon dioxide over time. Technicians must verify the exact molarity of the titrant by performing a standardized titration against a primary standard reference material, such as potassium hydrogen phthalate (KHP). Utilizing an unstandardized titrant directly invalidates the mathematical calculations performed by the titrator, rendering the final acidity profile completely useless for compliance purposes.
Furthermore, proper storage and physical maintenance of the equipment significantly extend the lifespan of the instrumentation. Electrodes must never be stored in distilled water, which aggressively leaches the internal electrolyte solution; instead, they must be submerged in a dedicated storage solution containing potassium chloride (KCl). Routine cleaning protocols utilizing enzymatic solutions are also required to strip away stubborn protein build-up that accumulates during the analysis of dairy and meat products.
The role of automated data integrity and audit trails in food laboratory titrators
Integrating automated data logging and secure audit trails into modern titration systems is essential for maintaining compliance with strict food safety data integrity standards, such as FDA 21 CFR Part 11. These advanced titrators automatically record every parameter of the analysis, including user identification, time stamps, raw electrode millivolt readings, and exact titrant consumption volumes, directly into secure databases. By eliminating manual transcription, laboratories prevent inadvertent data entry errors and protect against the intentional falsification of acidity profiles. This unalterable digital footprint guarantees that all analytical results used to verify the safety and quality of processed foods can be confidently defended during strict regulatory inspections and third-party quality audits.
Optimizing laboratory workflows for measuring acidity in processed foods
Securing analytical accuracy in food science with modern titrators requires a steadfast commitment to standardized chemistry principles and rigorous instrument maintenance. By understanding the distinct chemical differences between pH and titratable acidity, laboratories can provide a comprehensive safety and flavor profile for complex food matrices. Ultimately, evaluating acidity in processed foods with titrators safeguards public health, ensures strict regulatory compliance, and guarantees that global food manufacturers consistently deliver high-quality products to the consumer market.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.














