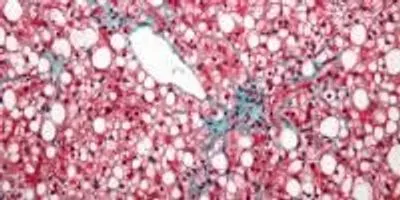
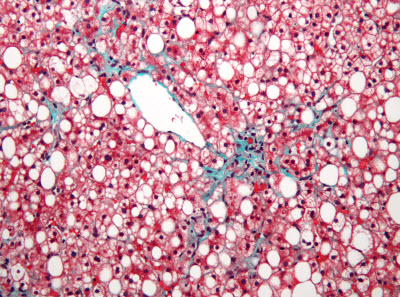
 Micrograph of non-alcoholic fatty liver disease (NAFLD).Image credit: Nephron, WIKIMEDIA COMMONSPHILADELPHIA— As obesity rates rise in the United States, so does the incidence of liver diseases. In fact, 80 percent of obese people are believed to have non-alcoholic fatty liver disease, or NAFLD, while another related liver disorder, non-alcoholic steatohepatitis (NASH), is expected to overtake hepatitis C as the leading cause of liver-related deaths, according to the National Institutes of Health. However, the mechanisms behind the toxic fat buildup linked to these diseases are not fully understood.
Micrograph of non-alcoholic fatty liver disease (NAFLD).Image credit: Nephron, WIKIMEDIA COMMONSPHILADELPHIA— As obesity rates rise in the United States, so does the incidence of liver diseases. In fact, 80 percent of obese people are believed to have non-alcoholic fatty liver disease, or NAFLD, while another related liver disorder, non-alcoholic steatohepatitis (NASH), is expected to overtake hepatitis C as the leading cause of liver-related deaths, according to the National Institutes of Health. However, the mechanisms behind the toxic fat buildup linked to these diseases are not fully understood.
Now, a new study published online Nov. 17 in Cell Metabolism by a team led by Mitchell Lazar, MD, PhD, director of the Institute for Diabetes, Obesity, and Metabolism in the Perelman School of Medicine at the University of Pennsylvania, revealed a surprising relationship between two molecules—one that works to store fat and another that promotes fat burning for energy. The team found that the molecules complement each other to maintain a healthy level of fat in the liver. When both molecules are removed from the liver, there is a swift buildup of toxic fats that causes an aggressive and lethal form of NASH in mice.
Related Article: Natural Sugar May Treat Fatty Liver Disease
The findings are an important discovery that could inform animal models for studying obesity and new drugs that reverse or prevent liver damage stemming from overeating.
When fat enters the body after a meal, it’s absorbed by the liver and either put away for safe storage in the form of triglycerides or it’s immediately metabolized for energy. The decision to burn or store fat depends on a number of factors, but the study found that the enzyme histone deacetylase 3 (HDAC3) and another protein SREBP are two critical regulators of this decision.
Earlier studies from the Lazar lab and others showed that disruption of either regulator causes problems in the liver: When HDAC3 is deleted experimentally from this system, fat piles up in the liver, but toxic fat is sequestered as triglycerides and therefore is relatively better-tolerated. Alternately, take SREBP away, and toxic fats get burned up and converted to energy.
Take both away, and “the results are catastrophic,” Lazar said. The mice had a dramatic inability to store triglycerides or burn excess fat safely in the liver.
“Based on what we first knew about these two molecules, we were expecting a tug of war that would fall somewhere in between if we took them both away, either too little fat or too much in the liver,” Lazar said. “Instead, it led to an accumulation of toxic fat and a complete energy drain on the mice that caused inflammation, liver damage, and ultimately death.”
The two molecules are back-up systems for each other, rather than a balance, as the team previously thought.
“It’s really two molecular pillars holding up a central ‘building’ to prevent liver toxicity,” he added. “This work shows that on a normal fat diet these two molecules hold down the fort in the mice.”
Taking away HDAC3 and SREBP in animal models could mirror the breakdown in the liver for investigators to study obesity and related liver diseases.
Related Article: Fatty Diets Lead to Daytime Sleepiness, Poor Sleep
“It’s more of a physiological discovery than explaining a disease; however, there are practical and conceptual implications here,” Lazar said. “These findings could serve as a model, in a short and acute way, of toxicity of fat in the liver that we can use to look for drugs that target these molecules to reverse or stop the damage.”
The study was led by Romeo Papazyan, a postdoctoral fellow in the Lazar lab, with help from Penn co-authors Zheng Sun, Yong Hoon Kim, Paul M. Titchenell, David A. Hill, Manashree Damle, Min Wan, Yuxiang Zhang, and Erika R. Briggs. Joshua D. Rabinowitz and Wenyun Lu of Princeton University are also authors.
The study was supported by the National Institutes of Health (R37 DK43806, F32 DK108555, R00 DK099443, F32 DK101175, T32 GM008216) and the JPB Foundation.