Repetitive strain injury (RSI) is one of the most prevalent occupational health risks in laboratory science, and manual pipetting is among its leading causes. Automated liquid handlers address this risk directly by removing the high-frequency, force-intensive, and posture-compromising movements that drive musculoskeletal damage — replacing human repetition with programmable, precision-controlled dispensing that benefits both staff wellbeing and data quality. For laboratory managers overseeing high-throughput workflows, understanding how these systems reduce injury risk is as important as understanding their throughput advantages.
Why manual pipetting puts laboratory workers at risk
The biomechanical demands of pipetting are easy to underestimate because each individual motion is small. The problem is cumulative exposure. A single aspiration and dispense cycle requires wrist deviation, sustained grip force, plunger depression, and tip ejection — four distinct physical actions — and laboratory technicians may complete hundreds or thousands of these cycles in a single shift.
Research published in the Scandinavian Journal of Work, Environment and Health found that pipetting for as few as 300 hours per year is associated with elevated rates of upper-extremity musculoskeletal disorders, including carpal tunnel syndrome, lateral epicondylitis, and thumb tenosynovitis.¹ NIOSH identifies repetition, force, and awkward posture as the three primary physical risk factors for work-related musculoskeletal disorders — all three are present in high-volume manual pipetting.² The situation compounds when scientists work in non-neutral postures: bent wrists during tip loading, elevated shoulders during extended reach, or sustained grip while aspirating viscous samples all accelerate tissue fatigue.
Tip ejection is a frequently overlooked contributor. The spring-loaded mechanism on many manual pipettes requires peak thumb force that exceeds recommended ergonomic thresholds, particularly for technicians with smaller hands. When multiplied across a 96-well plate workflow run dozens of times per week, that loading adds up to significant cumulative trauma.
How automated liquid handlers eliminate the injury pathway
Automated liquid handlers address RSI at the source by removing the human body from the physical execution of liquid transfer. A motorized pipetting head performs each aspiration, dispense, and tip ejection under software control — with consistent force, speed, and volume — while the operator's role shifts to loading, monitoring, and reviewing results.
This is more than a convenience improvement. When high-repetition manual pipetting steps are replaced with an automated workstation, the pipetting-related injury pathway is eliminated for those specific workflows. Staff can redirect physical effort toward tasks that require dexterity and judgment — instrument maintenance, sample receipt, data review — rather than sustaining repetitive mechanical motion across multi-hour sessions. Even partial automation of the highest-volume steps, such as reagent addition, serial dilution, or plate stamping, can meaningfully reduce daily exposure and give injured or at-risk staff a viable path to continued productivity.
Automation also eliminates the operator fatigue effect that compounds injury risk late in a shift. A liquid handler dispenses with identical precision at hour one and hour eight, whereas a fatiguing technician unconsciously modifies grip angle, tip depth, and aspiration speed — increasing both injury risk and pipetting error simultaneously.
Key ergonomic features to evaluate when selecting a system
Not all automated liquid handlers offer equivalent ergonomic design for the operator. Beyond pipetting performance specifications, laboratory managers should carefully evaluate the physical interaction demands that each instrument places on staff during setup, loading, and routine maintenance.
| Feature | Ergonomic benefit | What to verify |
|---|---|---|
| Motorized tip loading and pick-up | Eliminates manual tip-press force | Confirm tip pick-up force specification |
| Adjustable deck height | Supports neutral posture during loading | Check height adjustment range and locking stability |
| Touchscreen or remote programming | Reduces awkward arm position at terminal | Assess interface reach from a standing position |
| Automated tip ejection | Removes high-force thumb ejection impact | Request peak ejection force data |
| Covered or cassette-format consumable access | Minimizes repetitive bending for plate loading | Evaluate frequency of required manual plate intervention |
| Onboard barcode scanning | Reduces manual sample tube handling | Verify scanner speed and ergonomic position |
| Lightweight tip racks | Reduces strain during rack replacement | Weigh loaded racks and assess grip demands |
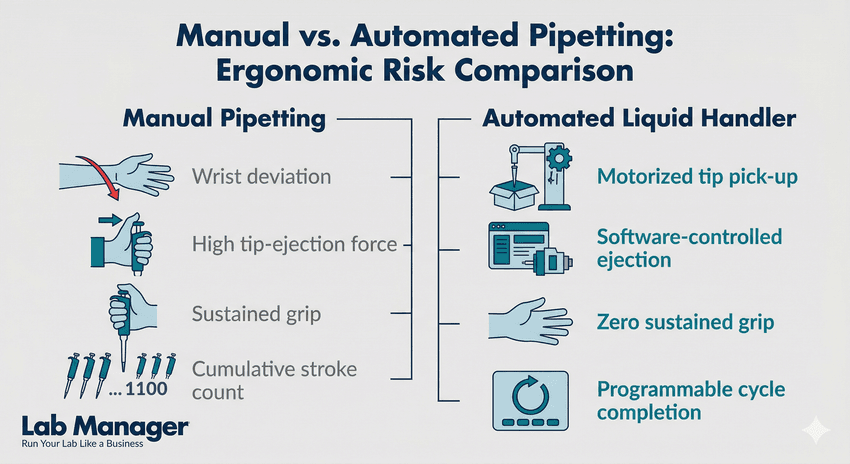
Repetitive strain isn't just a physical burden—it's a bottleneck for lab productivity. Our latest infographic breaks down the ergonomic shift from manual strain to automated precision. Learn how transitioning to automated liquid handlers can eliminate wrist deviation and sustained grip risks while boosting your throughput.
GEMINI (2026)
Software usability is also a genuine ergonomic factor that often goes unexamined. Systems that require complex programming at a fixed terminal, sustained periods of mouse-based navigation, or lengthy troubleshooting sessions create secondary ergonomic burdens that can partially offset the benefits gained from automating the pipetting itself. Intuitive graphical workflow editors and remote monitoring capability allow staff to interact with the system in postures of their choosing rather than in a fixed seated position at a dedicated workstation.
Building a transition plan that protects your team
Moving from manual to automated pipetting requires a structured implementation plan to maximize ergonomic benefit and minimize operational disruption. Rushing the transition — or only partially automating a workflow — can create new risks if operators are inadequately trained on instrument loading protocols or if high-frequency manual steps remain embedded in an otherwise automated process.
A practical transition framework includes the following:
- Audit current pipetting exposure. Identify the three to five workflows with the highest daily repetition counts and document how many manual pipette strokes each technician completes per shift. This baseline justifies capital investment and creates a benchmark for post-automation comparison.
- Prioritize complete workflow automation. Automate one workflow end-to-end before expanding to others. Partial automation that leaves high-frequency manual steps in place reduces return on investment and may not provide meaningful ergonomic relief.
- Evaluate residual manual tasks. After automation, audit remaining manual interactions — loading racks, uncapping tubes, handling plates — for accumulated residual strain. Ergonomic pipette selection and workstation height adjustment remain relevant for tasks that cannot yet be automated.
- Train on safe instrument interaction. Ensure all operators receive instruction not only on software and liquid handling protocols but also on safe loading positions, deck access technique, and correct body mechanics when handling consumable racks.
- Establish a reporting pathway for early symptoms. RSI responds far better to early intervention than to treatment after chronic injury has developed. A clear, non-punitive mechanism for staff to report discomfort early allows management to adjust workflows before injury occurs.
OSHA's ergonomics program principles recommend prioritizing engineering controls — structural changes to the work environment, including automation — as the most effective tier of intervention for preventing musculoskeletal disorders.² Implementing an automated liquid handler is applying the highest available ergonomic control for pipetting-intensive laboratory tasks.
For laboratories evaluating system options, Lab Manager's independent guide to purchasing an automated liquid handler provides a detailed breakdown of instrument types, throughput tiers, and procurement considerations. Ergonomic design should be evaluated alongside performance specifications during the selection process, not treated as a secondary factor.
Measuring the impact after implementation
Tracking the ergonomic outcome of automation is important both for validating the investment and for identifying any residual risk that requires additional intervention. Simple pre- and post-implementation metrics include self-reported discomfort scores using a standardized tool such as the Nordic Musculoskeletal Questionnaire, manual pipette stroke counts per technician per shift, and absenteeism rates attributable to musculoskeletal complaints.
Productivity metrics — throughput, plate error rates, and technician overtime — will often improve in parallel with ergonomic outcomes, reinforcing the dual business case for automation. Laboratories that document these outcomes create the evidence base needed to justify further investment in automation expansion or in complementary ergonomic interventions such as sit-stand workstations and anti-fatigue matting for residual manual tasks.
For broader context on managing musculoskeletal risk across all lab activities — not just pipetting — Lab Manager's ergonomic best practices guide covers posture, workstation setup, and break scheduling strategies applicable to the full range of laboratory workflows.
Protecting your team with automated liquid handling
Automated liquid handlers are among the most effective engineering controls available for reducing RSI risk in the research laboratory. By removing operators from repetitive, force-intensive pipetting tasks, these systems address the biomechanical root causes of upper-extremity injury while simultaneously improving throughput, reproducibility, and staff capacity. Selecting systems with ergonomic deck design, intuitive software, low-force consumable handling, and adjustable working height maximizes the health benefit beyond pipetting mechanics alone. Laboratory managers who treat automated liquid handling as both a productivity investment and an occupational health priority will see returns in both staff wellbeing and operational performance.
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.
References
- Björkstén, M. G., Almby, B., & Jansson, E. S. (1994). Hand and shoulder ailments among laboratory technicians using modern plunger-operated pipettes. Applied Ergonomics, 25(2), 88–94. https://doi.org/10.1016/0003-6870(94)90005-1
- NIOSH. (1997). Musculoskeletal Disorders and Workplace Factors: A Critical Review of Epidemiologic Evidence for Work-Related Musculoskeletal Disorders of the Neck, Upper Extremity, and Low Back. DHHS (NIOSH) Publication No. 97-141. https://www.cdc.gov/niosh/docs/97-141/default.html
- Rempel, D., Tittiranonda, P., Burastero, S., Hudes, M., & So, Y. (1999). Effect of keyboard keyswitch design on hand pain. Journal of Occupational and Environmental Medicine, 41(2), 111–119. (Comparative ergonomic intervention methodology reference)
















