WASHINGTON, D.C. – A survey published today (June 30) on research antibody usage and validation practices indicates many junior scientists are not receiving the training they need in the selection, handling, applications, and reporting of research antibodies. “The need for improved education and training in research antibody usage and validation practices” by Leonard P. Freedman, PhD, published in BioTechniques, describes a perception among all researchers that antibody validation delays, rather than facilitates research progress.
Research antibodies are indispensable biological reagents in basic and translational laboratories and clinical settings around the world. They are used in a wide variety of biomedical applications, with global sales of $1.6 billion in 2011 estimated to increase to $2.6 billion by 2019, yet they are a major contributor to the life science research reproducibility crisis.
Related Webinar: Selecting, Validating, and Publishing with Antibodies
The survey is first in a series of crowdsourcing strategies that also include an online discussion forum underway now and a workshop the Global Biological Standards Institute (GBSI) is hosting in September to build consensus and recommendations among the life science research community for improved accuracy in antibody usage and validation.
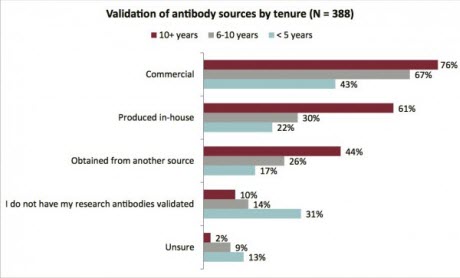 Figure 2. Validation of antibody sources by tenure.Image courtesy of GBSIResponses from more than 500 scientists within the life science community show that although 70 percent of respondents validate or have commercial antibodies validated, just 43 percent of researchers with fewer than five years of experience report validating purchased antibodies, and only 22 percent report validating antibodies produced in-house. Still more troubling is the finding that a third of these future principal investigators (PIs) do not validate at all (Figure 2). Only 44 percent of the respondents reported receiving specific training on the importance of validating, and how to validate research antibodies for specific applications.
Figure 2. Validation of antibody sources by tenure.Image courtesy of GBSIResponses from more than 500 scientists within the life science community show that although 70 percent of respondents validate or have commercial antibodies validated, just 43 percent of researchers with fewer than five years of experience report validating purchased antibodies, and only 22 percent report validating antibodies produced in-house. Still more troubling is the finding that a third of these future principal investigators (PIs) do not validate at all (Figure 2). Only 44 percent of the respondents reported receiving specific training on the importance of validating, and how to validate research antibodies for specific applications.
“GBSI’s Research Antibodies Survey clearly identifies a need to improve the skills and proficiency of junior scientists in application-specific antibody validation and to develop validation standards that are widely accepted by the research community,” said Leonard P. Freedman, PhD, president of GBSI. “The biomedical research community must commit sufficient time, resources, and expertise to educate and train scientists—particularly young researchers—in best practices for antibody-based experiments and reporting, and in the importance of application-specific validation.”
 Figure 3. Barriers to performing antibody validation by the respondents' years of antibody use.Image courtesy of GBSIWhen assessing barriers to validation of antibodies in their research, a clear trend emerges based on years of experience—more than one-fourth of researchers with fewer than five years of antibody use indicated that they “do not see the necessity” of performing antibody validation. Other perceived barriers, include “takes too much time” (71 percent) and “delays my research” (51 percent) (Figure 3).
Figure 3. Barriers to performing antibody validation by the respondents' years of antibody use.Image courtesy of GBSIWhen assessing barriers to validation of antibodies in their research, a clear trend emerges based on years of experience—more than one-fourth of researchers with fewer than five years of antibody use indicated that they “do not see the necessity” of performing antibody validation. Other perceived barriers, include “takes too much time” (71 percent) and “delays my research” (51 percent) (Figure 3).
“Despite prolific use, and the knowledge that research antibodies are a major contributor to the current reproducibility crisis, there are currently no standardized and widely accepted guidelines for antibody validation,” said Freedman.
Related Article: New Antibody Portal Bolsters Biomedical Research Reliability
“A variety of issues can jeopardize antibody-dependent experiments—including age, stability, inappropriate storage and handling, and lot-to-lot variation of antibodies—resulting in widespread lack of trust in antibody quality control and specificity. The inability to reproduce biomedical research can often be traced to false-positive or false-negative results produced with antibodies that were not properly validated for the intended use both on the part of the supplier and the user. And even when validation data is provided by a vendor, it is often inadequate, dated, and unreliable.”
The anonymous online survey generated responses from 504 scientists who use antibodies in their research—90 percent conduct basic research and 74 percent work in academia. Go to GBSI.org for full survey results.
Antibody validation workshop at Asilomar- Sept. 25-27
GBSI in conjunction with The Antibody Society will bring together key stakeholders from academia, antibody producers, pharma, funders, and journals to recommend standards for validating antibodies, September 25-27, 2016 at Asilomar Conference Grounds in Pacific Grove, California. Participation in the “Antibody Validation: Standards, Policies, and Practices” workshop is by invitation only. Space is limited, but GBSI is accepting requests from those who would like to attend and/or join in sponsoring the workshop. Sponsors currently include The National Institutes for Health,* The Antibody Society, Abcam, Cell Signaling Technology, Grifols, OriGene, and Rockland Immunochemicals.
Pre-workshop online discussion: Crowdsourcing Antibody Validation Solutions
GBSI invites participation in an online dialogue through August 15, engaging the scientific community to build consensus for research antibody validation standards. The GBSI Antibody Validation Online Discussion Group is a first-of-its-kind crowdsourcing approach that will inform the dialogue and outcomes at the September “Antibody Validation: Standards, Policies, and Practices” workshop. Online discussion topics include:
- Creating antibody standards: Establishing standards and validation techniques for biochemical and cell-based assays using antibody-based applications and methodologies
- Responsibilities of the companies: Engaging antibody suppliers to produce higher-quality, internally validated antibodies
- Drivers for Adoption: Ensuring that validation standards are accepted and practiced by the research community, in part through training and certification
- The Future of Antibodies: Integrating new technologies, including greater investment in recombinant antibodies
*Funding for the Antibody Validation workshop was made possible in part by an National Institutes of Health R-13 grant, GM 121024-01/G236SH. The views expressed in written conference materials or publications and by speakers and moderators do not necessarily reflect the official policies of the Department of Health and Human Services; nor does mention of trade names, commercial practices, or organizations imply endorsement by the U.S. Government.