Automated liquid handling integration is most likely to succeed when it is treated as a workflow redesign project, not simply a hardware installation. Labs that approach the process strategically — auditing existing workflows, identifying the highest-value integration points, and connecting the instrument to data systems before the first run — consistently achieve faster adoption and stronger returns than those that simply place a liquid handler on the bench and expect productivity gains to follow. For a broader overview of the technology and its capabilities, Lab Manager's complete guide to automated liquid handling provides essential context before tackling the operational integration steps covered here.
Audit your workflows before the instrument arrives
A structured pre-implementation audit is the foundation of any successful automated liquid handling integration. The objective is not to identify every manual step in the lab, but to locate the specific pipetting workflows that combine high repetition, large sample volume, and significant error risk — these are the candidates that will deliver the clearest return on investment.
Begin by documenting the five to ten most pipetting-intensive workflows in the lab, including the number of samples processed per run, the frequency of each workflow per week, and the average time a trained technician spends on each. Pair this with error data where available: failed assays, repeat runs, and CV (coefficient of variation) reports from manual pipetting steps often reveal the true cost of the status quo. Workflows with high stroke counts, narrow volume tolerances, or multi-reagent dispensing steps are the strongest candidates for automation.
The audit should also identify physical constraints. Available bench space, power supply locations, HEPA or biosafety cabinet requirements, and the proximity of reagent storage all affect instrument placement and deck configuration. Establishing these parameters before specifying a system avoids costly reconfigurations after delivery.
| Workflow assessment criteria | Why it matters |
|---|---|
| Daily pipette stroke count | Quantifies RSI risk and operator time burden |
| Coefficient of variation (CV) | Identifies assays where manual variability is a quality risk |
| Sample batch size | Determines whether throughput justifies automation |
| Regulatory documentation requirements | Flags workflows where audit trail capability is mandatory |
| Reagent sensitivity (temperature, light, evaporation) | Informs deck configuration and environmental controls |
Match the integration strategy to your throughput tier
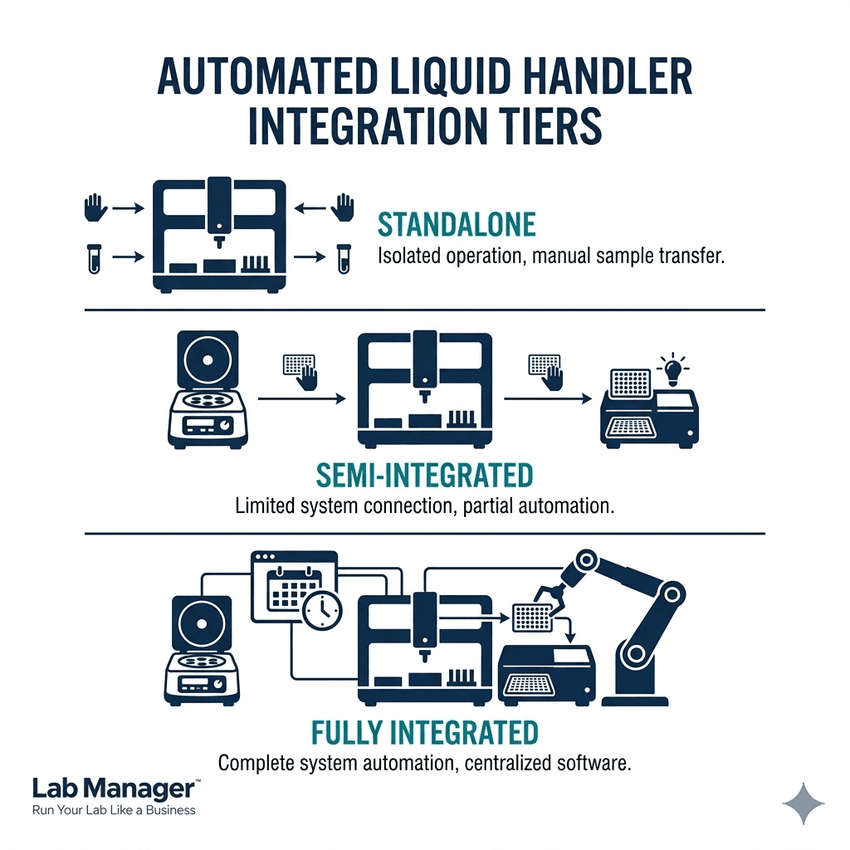
From standalone setups to fully automated workcells—which integration tier is your lab operating at? Level up your throughput with this quick guide to liquid handler automation.
GEMINI (2026)
Not all liquid handlers integrate into existing workflows in the same way, and the correct approach depends on both instrument type and the lab's operational model. A bulk reagent dispenser optimized for plate filling operates differently — and connects to surrounding workflows differently — than a full-workstation system capable of serial dilutions, reformatting, and cherry-picking across multiple plate formats.
For labs introducing their first liquid handler, a phased integration strategy reduces disruption. Automating one complete workflow end-to-end before expanding to additional applications gives operators time to develop programming competence and validates instrument performance within the lab's specific environment. Attempting to automate multiple workflows simultaneously is a common failure mode: it fragments training, divides troubleshooting attention, and produces no clean data set to benchmark against.
For higher-throughput environments, integration often involves scheduling the liquid handler as a shared resource across multiple workflows or teams. In these settings, a run queue — a managed schedule governing access and prioritizing urgent samples — is essential. Instrument utilization rates, deck changeover time, and tip consumption all need to be tracked to prevent the liquid handler from becoming a scheduling bottleneck rather than a productivity asset.
- Standalone operation: The liquid handler completes a self-contained step (e.g., plate replication) independently, with manual handoff before and after.
- Semi-integrated operation: The liquid handler is positioned inline with centrifuges, plate readers, or incubators, with manual transfer between stations.
- Fully integrated operation: The liquid handler is embedded within a robotic workcell or scheduling software platform that manages sample flow automatically across multiple instruments.
Laboratories evaluating which tier fits their operation should consult Lab Manager's independent guide to automated liquid handler procurement for a detailed breakdown of instrument classes and throughput capabilities before committing to a configuration.
Connect the liquid handler to your data infrastructure
Automated liquid handling integration delivers its full value only when instrument output connects directly to the lab's data management systems. A liquid handler that generates run logs in a proprietary format that must be manually re-entered into a Laboratory Information Management System (LIMS) or electronic lab notebook (ELN) reintroduces the transcription errors and audit trail gaps that automation is meant to eliminate.
Most modern liquid handlers support bidirectional data transfer via CSV export, direct LIMS API integration, or instrument-specific middleware. The appropriate path depends on the LIMS platform in use and the IT resources available to configure the connection. At a minimum, the instrument should be configured to automatically archive timestamped run logs — including operator ID, protocol version, volume dispensed per well, and any flagged errors — to a location accessible by the LIMS or ELN. As Lab Manager has reported, integrating LIMS and ERP systems can automate data handoffs across departments that would otherwise require manual coordination, and the same principle applies to instrument-level integration.
For regulated environments operating under FDA 21 CFR Part 11 or equivalent frameworks, audit trail completeness is non-negotiable. Each method change, operator login, and deviation from protocol must be captured without manual intervention. Verifying that the liquid handler's software architecture meets these requirements before purchase — not during validation — saves significant remediation effort.
Validate protocols and train staff concurrently
Instrument qualification and staff training are frequently treated as sequential activities: first the instrument is validated, then operators are trained. Running them in parallel — with experienced staff involved in the qualification runs themselves — accelerates adoption and produces better protocols. Operators who understand why a method was built the way it was are better equipped to troubleshoot it and less likely to revert to manual techniques when problems arise.
Protocol validation for liquid handling integration should include at a minimum: aspiration and dispense accuracy across the full volume range, inter-well CV across a full plate at the target volume, carryover testing between sample types, and tip engagement and ejection force verification for the consumable format in use. Documenting all validation runs with the same rigor as a method transfer creates a defensible baseline for future performance trending.
Staff transition also requires attention to the workflows that remain manual. Automating the core pipetting step in an assay while leaving reagent preparation, plate labeling, and centrifugation as manual tasks can create new bottlenecks at the handoff points. A brief time-and-motion study of the fully revised workflow — manual steps included — identifies these pinch points before they affect throughput. Labs navigating broader automation implementation challenges will find practical guidance in Lab Manager's article on securing buy-in and justifying ROI for lab automation.
It is also worth reviewing ergonomic exposure for residual manual tasks. Automated liquid handling integration substantially reduces pipetting-related musculoskeletal risk, as covered in detail in Lab Manager's analysis of RSI prevention through liquid handler adoption, but residual plate handling and rack loading should still be assessed for cumulative strain.
Measuring integration success and optimizing over time
Successful automated liquid handling integration is not a one-time event — it is an ongoing operational discipline. Establishing clear performance metrics from the outset allows lab managers to demonstrate value to leadership and identify degradation before it affects data quality.
Key metrics to track post-integration include instrument utilization rate (percentage of available capacity used), inter-run CV for core assays, consumable consumption per run, technician time freed from pipetting tasks, and the frequency of error flags or aborted runs. A liquid handler consistently operating below 40% utilization may indicate that workflows have not been fully transitioned to the system, or that the instrument's capacity exceeds current lab demand. One operating at near-100% utilization with frequent scheduling conflicts signals the opposite problem. Reviewing these metrics quarterly — rather than only when a problem emerges — allows proactive adjustment before operational impact occurs.
Method libraries also benefit from regular review. As new assay types are introduced or reagent formulations change, existing protocols may require recalibration. Maintaining a versioned, access-controlled protocol repository — ideally within the LIMS — ensures that operators are always running the current validated method and that changes are traceable. Automated liquid handling integration that is maintained this way creates a durable operational asset, not a system that gradually drifts from its validated state.
References
- U.S. Food and Drug Administration. (2003). 21 CFR Part 11: Electronic records; electronic signatures — scope and application. FDA Guidance for Industry. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/part-11-electronic-records-electronic-signatures-scope-and-application
- Tamburri, A., & Gamble, R. (2024, June 21). Planning for laboratory automation. LabCon 2024, Canadian Society for Medical Laboratory Science. https://labcon.csmls.org/wp-content/uploads/2024/08/Planning-for-Laboratory-Automation-Presentation-Labcon-2024V5.pdf
- U.S. Bureau of Labor Statistics. (2024). Occupational outlook handbook: Clinical laboratory technologists and technicians. https://www.bls.gov/ooh/healthcare/clinical-laboratory-technologists-and-technicians.htm
This article was created with the assistance of Generative AI and has undergone editorial review before publishing.
















